
زيادة مبيعات شركة حورس للأدوية
حققت شركة «حورس» للأدوية التابعة لإيفا فارما مبيعات بقيمة مليار مليار جنيه في عام 2021. وزادت مبيعات شركة حورس للصناعات الدوائية، في الربع الأول من العام الجاري 22022 بنسبة 37.4%، بقيمة 532.2 مليون جنيه، وحصة سوقية 2%، مقارنة بنفس الفترة من 2021. أكد منير ارمانيوس، رئيس مجلس إداره شركه حورس للصناعات الدوائيه، أن شركته تهدف لتلبيه رغبات واحتياجات المواطن المصري في توفير علاج فعال، خاصة أن المشروع مصنع حورس مصمم طبقًا لقواعد التصنيع الاوروبي GMP و يوفر 1100 فرصة عمل وينتج المصنع أدوية متطوره للاستهلاك المحلي والتصدير، على إجمالي مساحه 125 الف متر مربع . وأغلقت مجموعة إيفا فارما تطبيق بيع الدواء والمكملات الغذائية، وأصدرت بيانًا جاء فية: صيادلة مصر الأفاضل،،، وردت إلينا شكاوى عدد من الزملاء، حول بيع مستحضرات خاصة بالشركة عن طريق برنامج إلكتروني يتيح تقديم خصومات عبر التعامل المباشر مع الجمهور، وهو أمر غير صحيح على الإطلاق؛ حيث أن مجموعة شركات إيفا تضع نصب أعينها كلاً من مصلحة جموع المواطنين، والتخفيف عن كاهلهم، ولا يمكن أن نغفل بأي حال من الأحوال مصالح شركائها من صيادلة مصر. ونظرا لسوء التفاهم نوضح ، أن الواقعة تعود لبرنامج يجري العمل عليه بشكل تجريبي، ولم يتم إطلاق الخصومات سوى لتحقيق المصلحة المشتركة لكلاً من المواطن المصري والصيدلي؛ حيث يتم صرف…
حجم أعمال «فارما أوفرسيز» يتخطى 500 مليون جنيه
September 27, 2022 Comments Off on حجم أعمال «فارما أوفرسيز» يتخطى 500 مليون جنيه
قال الدكتور مهاب جزارين، رئيس مجلس إدارة شركة فارما أوفرسيز إن استثمارات شركته تتخطى 500 مليون جنيه للأصول الثابتة، وسلاسل الإمداد، والتطور التكنولوجي، موضحًا أنها تضم أكثر من 40 فرعًا، موزعة على كل محافظات الجمهورية. وأضاف فى حوار خاص لـ«المال»، أن الشركة تضم 5000 موظف، وهى موجودة منذ أكثر من 25 عامًا. وأكد أن «فارما أوفرسيز» تستهدف نموًا بنسبة %25 خلال العام المقبل، عبر استحداث أدوات التواصل الإلكترونية التى وفرتها الشركة للصيادلة. وأوضح اهتمام «فارما أوفرسيز» بالتحول الرقمي، فى ظل مميزات التطبيقات التكنولوجية فى التسويق، وتوفير الوقت للعميل والشركة فى آن واحد، لافتًا إلى أنها عملت على ميكنة كل المستندات الداخلية لكل المعاملات التجارية منذ بداية العام. وكشف عن التعامل مع هيئة الشراء الموحد، من خلال الفاتورة الإلكترونية، إذ ترفع بشكل رقمى على الموقع الخاص بمصلحة الضرائب ووزارة المالية مما يُسهل التعامل مع الأجهزة الحكومية. التطبيق الإلكترونى للصيادلة وأشار الدكتور مهاب جزارين إلى أن «فارما أوفرسيز» أول شركة توزيع دواء تقوم بتوفير تطبيق هواتف ذكية لخدمة عملائها من الصيادلة، وشرح آلية العمل، إذ يقوم الصيدلى بعمل كل احتياجاته من خلال الشركة بشكل رقمي، فى أى وقت. كما يمكنه متابعة حجم مسحوباته، وعمل طلبياته بشكل سهل وبسيط، والاطلاع على الأصناف المتوافرة للمنتجات المثيلة، إضافة إلى العروض المقدمة على تلك الأصناف، وعمل…
Continue reading …ديلان جاكسون رئيسًا لشركة “جلاكسو سميثكلاين مصر”
June 7, 2022 Comments Off on ديلان جاكسون رئيسًا لشركة “جلاكسو سميثكلاين مصر”
قامت شركة جلاكسو سميثكلاين بتعيين ديلان جاكسون رئيساً لمجلس إدارة الشركة ومحمد الضبابي عضواً منتدباً ومديراً تنفيذياً لها بمصر. وتم اختيار ديلان جاكسون رئيساً لمجلس الإدارة خلفا لأشرف منصور، والذي قاد خلال فترة رئاسته ، مشروعات وبرامج استراتيجية لجلاكسو سميثكلاين في السوق المصرية، أدت إلى نمو كبير في قطاع الصيدلة و صناعة الدواء وكان لها دوراً مؤثرا في جعل الأدوية و اللقاحات الأساسية في متناول شريحة أوسع من المصريين. قبل توليه رئاسة مجلس إدارة جلاكسو سميثكلاين مصر، تدرج جاكسون منذ عمله بجلاكسو سميث كلاين لمدة تزيد عن 10 أعوام، في العديد من المناصب القيادية والهامة بالشركة، استطاع خلالها توسيع حجم مبيعات وعمليات الشركة بأكثر من 60 سوقاً ناشئاً. وساهم في دعم وتنفيذ خطط الشركة للنفاذ إلى أسواق جديدة، وتطوير الشراكات مع الحكومات وأصحاب المصالح المختلفين، بالإضافة إلى قيادة عملية الدمج والاستحواذ على شركة نوفارتس في الأسواق الناشئة. وتمتد خبرة جاكسون في التسويق الطبي والتخطيط الاستراتيجي إلى أكثر من 23 عاماً، عمل خلالها استشارياً لدى شركات الدواء الكبرى في أسواق مختلفة حول العالم، كما ساهم في دعم المبيعات والتسويق لمنتجات عالمية ورائدة. ووقع الاختيار على محمد الضبابي كعضو منتدب ومدير تنفيذي ، وهو خبير متمرس لديه تاريخ طويل في مجال الصيدلة وصناعة الدواء، و يستند إلى خلفية قوية في الصناعة جاءت…
Continue reading …«فوربس» تختار رئيس «فاركو» ضمن أهم 50 قائدا لشركات الرعاية الصحية
April 11, 2022 Comments Off on «فوربس» تختار رئيس «فاركو» ضمن أهم 50 قائدا لشركات الرعاية الصحية
اختارت مجلة فوربس الشرق الأوسط، الدكتور شيرين حلمي الرئيس التنفيذي والعضو المنتدب لشركة فاركو للأدوية، فى المركز الـ12 فى الشرق الأوسط. والمركز الأول فى مصر ضمن قائمة أقوى 50 قائدا لشركات الرعاية الصحية في الشرق الأوسط، وهي قائمة تضم رؤساء أكبر مؤسسات القطاع الصحي الأكثر تأثيرًا على حياة ملايين الأفراد في المنطقة وخارجها. يأتي ذلك تتويجًا لمسيرته المهنية التي تمتد لأكثر من 30 عامًا في مجال الصناعات الدوائية، والتي تميزت بالعديد من الإنجازات على المستويات المحلية والإقليمية والدولية ساهمت في الارتقاء بقطاع الصناعات الدوائية. وبناءً على تلك المعطيات تصدر الدكتور شيرين حلمي الشركات المصرية بالقائمة، وتفوق على مؤسسات وكيانات كبرى تعمل بقطاع الرعاية الصحية في العديد من الدول العربية. يأتي ذلك بفضل نجاح “حلمي” في قيادة مجموعة فاركو منذ تأسيسها عام 1983 في تطوير وتصنيع وتوزيع الأدوية لأكثر من 50 دولة، علاوة على قيادة الشركة في المساهمة في القضاء على التهاب الكبد الوبائي في مصر، وتصنيع أدوية ولقاحات لعلاج والوقاية من فيروس كورونا. كما يساهم الدكتور شيرين حلمي في العمل العام من خلال عضوية العديد من مجالس الأعمال منها مجالس الأعمال المصرية اللبنانية والمصرية السعودية والسنغالية المصرية، بالإضافة إلى غرفة التجارة الأمريكية في مصر، والغرفة الألمانية العربية للصناعة والتجارة، وجمعية رجال الأعمال المصرية السويسرية، وغرفة التجارة والصناعة الفرنسية في مصر،…
Continue reading …” ايبيكو” تبدأ في تصنيع المواد الخام للادوية
February 21, 2022 Comments Off on ” ايبيكو” تبدأ في تصنيع المواد الخام للادوية
كشف د.حاتم البدوي سكرتير عام شعبة الصيدليات باتحاد الغرف التجارية عن قيام شركة ايبيكو ببدء خطوات فعلية لتصنيع المادة الخام وراى انها خطوة هامة من شركة وطنية مصرية واكد انها ستكون خطوة حيوية في صناعة الدواء . واوضح ان مصر لديها كل مقومات نجاح صناعة الدواء ومقومات التصدير لكل الاسواق . وقال ” لسنا اقل من دول مثل اسرائيل التي تصدر ب 7 مليار في السنة والاردن التي تصدر بنحو 4 مليار في السنة ” . في الوقت نفسه راى ان التسعير الجبري من اهم المشاكل التي تقابل الصيادلة لانه يؤدي الى مشكلة عند تسجيل الدواء في دولة اخرى . واستطرد ” لايستطيع الصيدلي ان يبيع الدواء بخلاف السعر المكتوب عليه وحتى بعد الزيادات لا يصل سعر الدواء الى تكلفته . واضاف ” لا يوجد دواء في العالم ثمنه 2 جنيه او 3 جنيه وهو مايجعل الشركات تمتنع عن تصنيعه لعدم تغطية تكلفته والحل في تطبيق منظومة التامين الشامل وأرى ان الدواء المحلي الصنع اكثر امانا من الدواء المستورد المهرب ” .
Continue reading …“استرازينيكا مصر للأدوية” تخطط لمضاعفة استثماراتها خلال 3 سنوات
February 19, 2022 Comments Off on “استرازينيكا مصر للأدوية” تخطط لمضاعفة استثماراتها خلال 3 سنوات
تخطط شركة أسترازينيكا مصر للأدوية مضاعفة استثماراتها فى السوق المصرى خلال السنوات الثلاثة المقبلة . وقال حاتم الوردانى رئيس مجلس إدارة الشركة إن أسترازينيكا مصر تخطط لطرح 20 دواء جديدا بنهاية 2025، وسوق الدواء المصرى من الأسواق الواعدة وضمن الأكثر نموا فى الشرق الأوسط وأفريقيا واسترازينيكا تعمل على دعم قطاع الرعاية الصحية فى مصر. أوضح أن الشركة تخطط لتدريب 1000 صيدلى مصرى على التكنولوجيا الصحية خلال العام الجارى بالتعاون مع كلية لندن للاقتصاد. وحققت شركة أسترازينيكا مصر للأدوية مبيعات بقيمة 1.923 مليار جنيه خلال العام الماضى بنسبة نمو 15.8 % وتعمل الشركة فى السوق المصرى منذ 1968 وتمتلك مصنعا بمدينة السادس من أكتوبر منذ عام 2006 على مساحة 6500 متر مربع بطاقة إنتاجية تصل إلى 900 مليون قرص سنويا. ووقعت أسترازينيكا للأدوية الأسبوع الماضى بروتوكول تعاون مع الهيئة المصرية للشراء الموحد والإمداد والتموين الطبي مذكرة تفاهم لرعاية المؤتمر الافريقي المزمع انعقاده منتصف العام الجارى. وتهدف المذكرة لتعزيز العلاقات التجارية بين الهيئة وأسترازينيكا وتبادل الخبرات في مجال الأعمال والاستثمارات في قطاع الرعاية الصحية مع استمرارية التعاون للعام الثاني لتدريب الصيادلة على التعامل الكامل مع مستحدثات، وأخر تطورات التكنولوجيا الصحية في العالم. أضاف أن مذكرة التفاهم تأتى تنفيذا لاستراتيجية أسترازينيكا التى تهدف لدعم وتنمية وتطوير القطاع الصحي في مصر، بكافة السبل والإمكانيات…
Continue reading …«ايفا فارما» تحتل مركز الوصيف بين شركات الأدوية الأعلى مبيعا في مايو.. وتنمو بنسبة 49%
July 6, 2021 Comments Off on «ايفا فارما» تحتل مركز الوصيف بين شركات الأدوية الأعلى مبيعا في مايو.. وتنمو بنسبة 49%
كشفت مصادر دوائية مطلعة عن نمو حجم مبيعات شركة ايفا فارما، خلال شهر مايو الماضي من خلال «الصيدليات» بنسبة 48.7%.، بقيمة مبيعات بلغت نحو 387.6 مليون جنيه، وحصة سوقية 6%،. لتحتل مركز الوصيف بين شركات الأدوية الأعلى مبيعا في مصر. وقالت المصادر في تصريحات خاصة لـ «سوق الدواء»، أن «ايفا فارما»، حققت مبيعات خلال الشهور الخمسة الأولى من 2021 بقيمة 1.559 مليار جنيه بنسبة نمو 26% وحصة سوقية 4.6% . ونوهت المصادر الى تحقيق «ايفا فارما»، مبيعات خلال الفترة من مايو 2020 الى مايو 2021، بقيمة 3.920 مليار جنيه، بنسبة نمو 32.% خلال نفس الفترة من العام السابق عليه، وحصة سوقية 4.76.6%. حجم مبيعات سوق الدواء المصري وارتفعت مبيعات سوق الدواء المصري في شهر مايو 2021، من خلال «القطاع الخاص» بنسبة 19.9% محققا حجم مبيعات بقيمة 6.440 مليار جنيه. وحقق سوق الدواء المصري مبيعات خلال الشهور الخمسة الأولى من 2021 بقيمة 32.5 مليار جنيه، بنسبة نمو 9.1%، بينما حقق سوق الدواء مبيعات خلال الفترة من مايو 2020 الى مايو2021، بقيمة 83.2 مليار جنيه بنمو بلغت نسبته 9.6%. أكبر 15 شركة دواء في مصر وأشارت المصادر الى تغيرات جوهرية في ترتيب مبيعات شركات الأدوية الـ 15 الكبار، حيث تصدرت شركات نوفارتس، ايفا فارما، آمون، سانوفي، ايبيكو، جلاكسو سميثكلاين، فاركو، فايزر، المهن…
Continue reading …شركة جونسون تقوم بإيقاف تصنيع مستحضرات تفتيح البشرة
June 22, 2020 Comments Off on شركة جونسون تقوم بإيقاف تصنيع مستحضرات تفتيح البشرة
قامت شركة جونسون آند جونسون الأمريكية بوقف تصنيع مستحضرات تفتيح البشرة وإزالة الرؤوس السوداء والبقع السوداء من الوجه، وذلك بعد الهجوم الإعلامي الكبير حول موضوع عدم المساواة العرقية التي حدثت مؤخراََ بعد حادثة مقتل الأمريكي من الأصول الأفريقية جورج فلويد الشهيرة. حيث بين المتحدث الرسمي للشركة أن شركة جونسون آند جونسون سوف تقوم بوقف خط الانتاج لمستحضر كلين آند كلير الذي يباع في الهند وأنه من الممكن أن تقوم الشركة بإيقاف خط انتاج مستحضرات نيتروجينا لتفتيح البشرة أيضاََ والذي يباع بشكل كبير في آسيا والشرق الأوسط. وأضاف المتحدث الرسمي باسم الشركة أنه خلال الأسابيع السابقة تم تداول الكثير من التعليقات بخصوص مستحضرات تفتيح البشرة المنتجة من الشركة وأن هناك إدعاءات أن الشركة تدعو لاستخدامها لأنها تقوم بتفتيح البشرة وتبييضها بشكل أجمل من لون البشرة الخاص والطبيعي لكل إنسان، حيث أوضح أيضاََ أن هذه لم تكن نية الشركة أباََ عند إنتاج مثل هذه المستحضرات وأن البشرة الجميلة والمميزة في البشرة الصحية والسليمة بغض النظر عن لونها. وأوضحت الشركة أيضاََ أنها سوف تقوم بإيقاف انتاج وتصنيع جميع هذه المستحضرات ولكم من الممكن تواجدها في الأسواق وذلك بسبب عدم انتهاء السلع المتواجدة حالياََ فيها. وتعتبر شركة جونسون آند جونسون الأمريكية إحدى أكبر الشركات المهتمة بتصنيع مستحضرات خاصة للبشرة والتي تقوم بتصنيع ما يزيد عن 6 آلاف طن من مستحضرات تفتيح البشرة والتي تحارب علامات تقدم السن مثل البقع…
Continue reading …روش للأدوية تبرم اتفاقية بقيمة 1.1 مليار دولار لتسويق علاج ضمور العضلات «دوشين» خارج أمريكا
December 24, 2019 Comments Off on روش للأدوية تبرم اتفاقية بقيمة 1.1 مليار دولار لتسويق علاج ضمور العضلات «دوشين» خارج أمريكا
أبرمت شركة روش للأدوية اتفاقية ترخيص بقيمة 1.15 مليار دولار مع شركة ساريبتا الأمريكية للأبحاث الطبية وتطوير الأدوية للحصول على حق في تسويق العلاج الجيني لشركة «ساريبتا» لعلاج ضمور العضلات «دوشين» خارج الولايات المتحدة. وقالت شركة الأدوية السويسرية أنها ستدفع 750 مليون دولار نقدا ونحو 400 مليون دولار بعد الإنتهاء من العلاج الجيني الدقيق الذي تطوره شركة «ساريبتا» حاليا وفقا لرويترز. وأضافت «روش» أن شركة «ساريبتا» ستكون مؤهلة من تلك الصفقة للحصول على مايصل الى 1.7 مليار دولار من صافي المبيعات، مشيرة الى أنه من المتوقع إغلاق الاتفاقية في الربع الأول من عام 2020. وأوضحت «روش» أن شركة «ساريبتا» ستظل هى المسئولة عن التطوير والتصنيع مع تقاسم تكاليف التطوير السريري بين الشركتين . جدير بالذكر أن ضمور العضلات «دوشين» هو اضطراب عضلي تنكسي نادر، يصيب حوالي واحد من بين 3500-5000 مولود ذكر في جميع أنحاء العالم ويسبب فقدان عضلي تدريجي حاد ووفيات مبكرة. 24 – December – 2019
Continue reading …«سيجما للأدوية» تستهدف مبيعات بقيمة 850 مليون جنيه 2020 وتطرح 11 مستحضرًا جديدًا
December 6, 2019 Comments Off on «سيجما للأدوية» تستهدف مبيعات بقيمة 850 مليون جنيه 2020 وتطرح 11 مستحضرًا جديدًا
كشف الدكتور عاطف دراز نائب رئيس مجلس الإدارة والعضو المنتدب للقطاع التجاري بشركة سيجما للادوية عن استراتيجية شركته خلال العام المقبل 2020 والتي يستهدف من خلالها التوسع بقوة داخل سوق الدواء المصري ومضاعفة حصة «سيجما» السوقية . وأضاف في تصريحاته أن استراتيجية شركته التوسعية تَرتَكز على العديد من المحاور أبرزها مُضاعفة حجم مبيعات الشركة لتتراوح بين 800 الى 850 مليون جنيه خلال 2020 لتصبح واحدة من الشركات الـ 30 الأكثر مبيعا في سوق الدواء المصري. وأشار الى إعتزام الشركة طرح 11 مستحضرًا جديدًا خلال العام المقبل، وذلك تماشيا مع خطتها الهادفة الى تصنيع وتوفير الأصناف الدوائية الحيوية لتلبية احتياجات ومتطلبات المرضى من خلال توفير الدواء الفعّال والآمن والذى يغطى كافه التخصصات الطبية والعلاجية ويتميز بالجودة العالية والاسعار المناسبة لكافه فئات المجتمع المصرى . وأوضح أن المستحضرات التى تعتزم «سيجما» طرحها خلال 2020 تتمثل في، مضادرات حيوية لعلاج، أمراض عدوى الجهاز التنفسى، وأمراض التهابات المسالك البولية، والوقاية من العدوى بعد العمليات الجراحية وكذلك الأمراض الجلدية المعدية وعلاج الحروق، الى جانب علاج تسمم الدم. وتابع : من بين المستحضرات التي تعتزم الشركة طرحها أيضا أدوية لعلاج أمراض الجهاز الهضمى والكبد، مثل إرتجاع المرئ، وحموضة المعدة وقرحة المعدة والاثنى عشر، وعلاج جرثومـة المعدة والقضاء عليها، والتهابات المرارة وعلاج الحصوات المرارية. بالإضافة الى أدوية لمنـع القئ والغثيان فى…
Continue reading …After CEO move, GSK picks ex-Novartis exec to head consumer unit
October 4, 2016 Comments Off on After CEO move, GSK picks ex-Novartis exec to head consumer unit
GlaxoSmithKline has promoted a former Novartis executive to lead its consumer healthcare business, following the appointment last week of the unit’s previous head, Emma Walmsley, as CEO designate of the drugmaker. The British group said on Thursday that Brian McNamara, 50, who currently heads up Europe and the Americas at GSK Consumer Healthcare, would take on his new role immediately and join GSK’s corporate executive team. McNamara worked for 11 years at Novartis, where he was also in charge of consumer health, and joined GSK following the creation of a joint venture between the two companies in 2015. Prior to that he spent 16 years at Procter & Gamble . The swift management transition will ensure the consumer business remains focused on improving financial performance and will allow Walmsley to focus on her new role as incoming CEO, a spokesman said. She will officially take the helm at the end of March on the retirement of current CEO Andrew Witty, becoming the first woman to lead a top global drugmaker. Separately, GSK said it had disposed of the final portion of its stake in Aspen Pharmacare , Africa’s biggest generic drugmaker, after seven years as an investor in the company….
Continue reading …باير الألمانية تستحوذ على مونستانو الأمريكية في صفقة ضخمة
September 19, 2016 Comments Off on باير الألمانية تستحوذ على مونستانو الأمريكية في صفقة ضخمة
كشفت شركة باير الألمانية للمستحضرات الطبية أن شركة شركة “مونسانتو” الامريكية للمبيدات وبذور المحاصيل قبلت عرض استحواذ بقيمة 66 مليار دولار، وبموجب هذه الصفقة تنشأ أكبر شركة في العالم للبذور والمبيدات الحشرية. ذكرت شركة “باير.إيه.جي” الألمانية العملاقة للمستحضرات الطبية أن شركة “مونسانتو” الامريكية للمبيدات الحشرية وبذور المحاصيل قبلت عرض الاستحواذ الذي قدمته. وفي بيان، قالت شركة “باير” إن “مونسانتو قبلت عرضها البالغ 128 دولارا لكل سهم، والذي يمثل قيمة إجمالية قدرها 66 مليار دولار. وكانت شركة “مونسانتو” قد رفضت ثلاثة اقتراحات سابقة للاستحواذ قدمتها شركة “باير” لان قيمة المبلغ كانت ضئيلة للغاية. وبموجب هذه الصفقة ستنشأ أكبر شركة في العالم للبذور والمبيدات الحشرية. وقال هيو جرانت، الرئيس التنفيذي لشركة /مونسانتو/ “نعتقد أن هذا الاندماج مع باير يمثل القيمة الأكثر إلحاحا لأصحاب الأسهم”. ومازالت الصفقة تستلزم موافقة الجهات الرقابية. 19 – September – 2016
Continue reading …$372M deal: GSK sells anaesthesia product line
September 19, 2016 Comments Off on $372M deal: GSK sells anaesthesia product line
GlaxoSmithKline entered into a deal Monday to sell its portfolio on anaesthesia products to Aspen Pharmacare Holdings for $239 million. The deal also includes milestone payments to GlaxoSmithKline (NYSE: GSK) of up to $133 million — putting the total value of the deal at up to $372 million. As part of the deal, the two companies also agreed to terminate their product marketing collaboration in Sub-Saharan Africa, and for Aspen to exercise its option to acquire GlaxoSmithKline’s remaining thrombosis business in China, India and Pakistan. Jefferson, Philadelphia U sign binding agreement to merge GSK’s anaesthesia portfolio consists of Ultiva, Nimbex, Tracrium, Mivacron and Anectine. The company, which is based in London and has large operations in Philadelphia and Montgomery County, previously divested the product line in the United States and Canada so it no longer sells the products in either country. Officials at GlaxoSmithkline said the agreement to sell the non-core assets is part of the company’s goal to focus on core therapeutic areas, which include cancer, metabolic and cardiovascular disease, inflammation, infectious diseases, neurosciences and vaccines. Aspen, which is based in South Africa, bought a portfolio on anaesthesia products from AstraZeneca (NYSE: AZN) in June in a deal valued…
Continue reading …Johnson & Johnson buys Abbott Medical Optics for $4.3B
September 19, 2016 Comments Off on Johnson & Johnson buys Abbott Medical Optics for $4.3B
Johnson & Johnson (JNJ) said Friday it is buyingAbbott Medical Optics, an eye-care unit of Abbott Laboratories (ABT), for $4.3 billion in cash. The deal adds additional ophthalmic products to Johnson & Johnson’s lineup including cataract surgery, laser refractive surgery and consumer eye health. J&J is one of the largest players in the global health care market with a variety of products including consumer products like contact lenses, pharmaceuticals and medical devices. “Eye health is one of the largest, fastest growing and most underserved segments in health care today,” says Ashley McEvoy, company group chairman at Johnson & Johnson’s vision care unit in a statement. “With the acquisition of Abbott Medical Optics’ strong and differentiated surgical ophthalmic portfolio, coupled with our world-leading … contact lens business, we will become a more broad-based leader in vision care. Importantly, with this acquisition we will enter cataract surgery – one of the most commonly performed surgeries and the number one cause of preventable blindness.” This is J&J’s only eyecare deal this year, says S&P Global Market Intelligence. It’s the largest deal with a disclosed amount since its $21 billion buy of Synthes in June, 2012 , which was announced in April, 2011, says S&P Global Market Intelligence….
Continue reading …Pfizer bolsters oncology portfolio with $14-billion deal to buy Medivation
August 25, 2016 Comments Off on Pfizer bolsters oncology portfolio with $14-billion deal to buy Medivation
Pfizer announced Monday a definitive merger agreement to acquire Medivation for $81.50 per share in cash, or approximately $14 billion, boosting its oncology portfolio with the addition of the prostate cancer therapy Xtandi (enzalutamide). The deal, which represents a 21-percent premium to Medivation’s closing share price on August 19 and has been approved by both companies’ boards of directors, is expected to complete in the third or fourth quarter. Pfizer CEO Ian Read said the purchase “is expected to immediately accelerate revenue growth and drive overall earnings growth potential.” The executive added that the transaction “will strengthen Pfizer’s Innovative Health business and accelerate its pathway to a leadership position in oncology, one of our key focus areas.” Xtandi generated around $2.2 billion in global sales over the past four quarters, as recorded by partner Astellas, with analysts estimating that the androgen receptor inhibitor could generate revenue of $5.7 billion by 2020. Meanwhile, Medivation’s pipeline includes the PARP inhibitor talazoparib, which is currently being investigated in a Phase III study for the treatment of BRCA-mutated breast cancer, and the immuno-oncology agent pidilizumab, which is being developed for diffuse large B-cell lymphoma and other haematologic malignancies. Albert Bourla, group president of Pfizer’s…
Continue reading …Pfizer will buy most of AstraZeneca’s antibiotics business in a $1.5bn (€1.32bn) deal that will see the Anglo-Swedish drug firm continue to package Merrem at its Macclesfield, UK plant for at least the next 18 months.
August 25, 2016 Comments Off on Pfizer will buy most of AstraZeneca’s antibiotics business in a $1.5bn (€1.32bn) deal that will see the Anglo-Swedish drug firm continue to package Merrem at its Macclesfield, UK plant for at least the next 18 months.
Under the terms of the agreement, Pfizer will pay $550m upfront and a further payment of $175m in 2019 for commercialisation and development rights to the late-stage antibiotics business. The US drug firm will also pay up to $250m in commercial, manufacturing and regulatory milestones, up to $600 million in sales-related payments as well as recurring, double-digit royalties on sales of Zavicefta and ATM-AVI in certain markets. The deal will not involve AstraZeneca’s biological anti-infectives, or the early-stage antibiotics business it established in 2015. Drugs In terms of products, AstraZeneca’s antibiotics business is a mixture of approved drugs and promising candidates. It sells Sumitomo Dainippon Pharma’s Merrem/Meronem (meropenem) outside certain Asian countries. It markets the cephalosporin Zinforo (ceftaroline fosamil) outside North America and Japan. Another drug in the portfolio, the combination antibiotic Zavicefta (ceftazidime-avibactam), was approved by the European Medicines Agency (EMA) in June. Candidate products include ATM-AVI, an injectable combination of aztreonam and a β-lactamase inhibitor, and CXL, an injectable combination of ceftaroline fosamil and Avibactam, both of which are being developed in collaboration with Allergan. Manufacturing AstraZeneca outsources production of its antibiotics portfolio. A spokesman for the firm told us that manufacturing contracts – which see contract manufacturing organisations (CMOs) in the…
Continue reading …Bristol-Myers Plunges On Surprise Opdivo Failure In Lung Cancer
August 7, 2016 Comments Off on Bristol-Myers Plunges On Surprise Opdivo Failure In Lung Cancer
Bristol-Myers Squibb (BMY) shocked Wall Street by announcing Friday that its new cancer drug Opdivo had failed a trial as a single therapy for newly diagnosed lung cancer patients. The company released few details of the study, called Checkmate-026, but it said that it hadn’t met its primary endpoint of improvement in progression-free survival in patients with advanced non-small-cell lung cancer (NSCLC). It was the first conspicuous miss for the drug since it launched in late 2014; it’s already been approved for six different indications in melanoma, kidney cancer, lymphoma and later-stage lung cancer after acing clinical trials. Bristol-Myers also reported positive data for Opdivo in front-line lung cancer in combination with another cancer drug, Yervoy, back in June. The Checkmate-026 failure is good news for Merck (MRK), whose competing drug Keytrudasucceeded in a similar trial recently. However, Merck played it safer in the patient population it chose, targeting “high expressers” of the PD-L1 ligand, which both Keytruda and Opdivo target through inhibition of the PD-1 protein in cancer cells. Bristol-Myers went for a broader patient population, potentially winning a bigger market but increasing its risk of failure. Bristol-Myers stock plunged 16% to 63.28 at the close on thestock market today, hitting a closing…
Continue reading …FDA Approves Adlyxin Sanofi Receives FDA Approval of Adlyxin (lixisenatide) for Treatment of Adults With Type 2 Diabetes
July 31, 2016 Comments Off on FDA Approves Adlyxin Sanofi Receives FDA Approval of Adlyxin (lixisenatide) for Treatment of Adults With Type 2 Diabetes
Sanofi announced today that the U.S. Food and Drug Administration (FDA) approved Adlyxin (lixisenatide), a once-daily mealtime GLP-1 receptor agonist injection indicated as an adjunct to diet and exercise for the treatment of adults with type 2 diabetes “The approval of Adlyxin reaffirms our continued commitment to addressing the challenges faced by people living with diabetes when trying to reach and maintain their individual blood glucose (HbA1c) targets,” said Peter Guenter, Executive Vice President, Head, Global Diabetes & Cardiovascular Business Unit, Sanofi. “We are pleased with this approval, as it offers us the opportunity to continue helping patients treated with basal insulin who remain uncontrolled.” The approval of Adlyxin was based on FDA review of results from the GetGoal clinical program and findings from the ELIXA trial, which successfully addressed the FDA’s request to demonstrate CV safety. The GetGoal clinical program, which included 13 clinical trials involving more than 5,000 adults with type 2 diabetes worldwide, evaluated the safety and efficacy of lixisenatide in adults with type 2 diabetes. All studies of the GetGoal program successfully met the primary efficacy endpoint of HbA1c reduction. The most common adverse events reported for Adlyxin included nausea, hypoglycemia and vomiting. Adlyxin will be…
Continue reading …Judge refuses to block generic versions of AstraZeneca’s Crestor
July 22, 2016 Comments Off on Judge refuses to block generic versions of AstraZeneca’s Crestor
A U.S. judge on Tuesday refused to issue a temporary restraining order blocking the approval of new generic versions of AstraZeneca’s blockbuster cholesterol drug Crestor in the United States. U.S. District Judge Randolph Moss in Washington, D.C. ruled that AstraZeneca was not likely to win a lawsuit claiming it should get seven more years of exclusive rights to the drug thanks to its recent approval to treat a rare pediatric illness. Generic drugmakers including Novartis AG unit Sandoz, Apotex and Mylan have said in court filings that they are prepared to launch generic versions of Crestor, which lost patent protection earlier this month. AstraZeneca spokeswoman Michele Meixell said in an emailed statement the company was “disappointed” with the decision. AstraZeneca’s bid to extend its exclusivity had drawn sharp criticism from former presidential candidate and Senator Bernie Sanders, a Democrat from Vermont, who along with seven Democratic U.S. Representatives had urged the U.S. Food and Drug Administration in a July 7 letter not to let the company exploit a “loophole.” Allergan plc has already been selling generic Crestor in the U.S. since May under a licensing agreement with AstraZeneca, and is not affected by Tuesday’s order. Crestor, which accounted for more…
Continue reading …«جلاكسوسميث كلاين» البريطانية تعتزم ضخ 340 مليون جنيه بقطاع الدواء المصرى
July 10, 2016 Comments Off on «جلاكسوسميث كلاين» البريطانية تعتزم ضخ 340 مليون جنيه بقطاع الدواء المصرى
تعتزم شركة جلاكسوسميث كلاين البريطانية للدواء، ضخ 340 مليون جنيه فى مصر حتى عام 2017، وفقا لما أكده عمرو ممدوح نائب الرئيس التنفيذى ومدير عمليات الشركة فى مصر، خلال لقاء جمعه بوزيرة الاستثمار داليا خورشيد، الاثنين. ووجهت الشركة دعوة لخورشيد لافتتاح خط انتاج جديد بمصنعها فى القاهرة خلال منتصف شهر سبتمبر القادم بتكلفة استثمارية تبلغ 60 مليون جنيه. وبحسب مسئولى الشركة فإن الخط الجديد بمصنع القاهرة سيقوم بإنتاج الأدوية الأساسية بأسعار فى متناول اليد، ضمن مبادرة جلاكسوسميث كلاين العالمية التى تهدف إلى ضمان وصول الدواء للمرضى من مختلف شرائح المجتمع. ويعد مصنعا القاهرة والجيزة التى تمتلكهما جلاكسوسميث كلاين فى مصر من أكبر مصانعها فى الشرق الاوسط وشمال افريقيا، حيث تقوم بتصدير 15 % على الأقل من إجمالى انتاج هذه المصانع إلى أكثر من 12 سوقا عالميا. 10 – July – 2016
Continue reading …AstraZeneca sells two dermatology drug rights to LEO Pharma
July 10, 2016 Comments Off on AstraZeneca sells two dermatology drug rights to LEO Pharma
British drugmaker AstraZeneca Plc said it sold the rights to develop an experimental mid-stage drug to treat atopic dermatitis to privately-held LEO Pharma. LEO Pharma would pay AstraZeneca $115 million in upfront payment and $1 billion in milestones. However, AstraZeneca will retain rights to develop the drug, tralokinumab, to treat severe asthma, the company said. Shares of AstraZeneca were trading nearly flat at 4,463 pence on Friday at 7.04 GMT on the London Stock Exchange. Separately, AstraZeneca said it terminated its licensing deal with Canadian drugmaker Valeant Pharmaceuticals International Inc on its experimental psoriasis drug, brodalumab, and sold the rights to that drug in Europe to LEO Pharma. Details of the deal were not disclosed. AstraZeneca said it did not expect the deals to have any impact on its earnings forecast. AstraZeneca’s deal with LEO Pharma falls in line with the British drugmaker’s recent strategy of focusing on cancer treatments and keeping its market position in developing respiratory treatments. AstraZeneca, which badly needs new drugs to combat the loss of patent protection on its older drugs, is pinning its hopes on another experimental treatment for severe asthma, benralizumab, while it develops more than 10 other respiratory treatments. In June alone,…
Continue reading …Sanofi has agreed to exchange its Merial animal health business for Boehringer Ingelheim’s consumer healthcare unit in a deal that will see €21.8bn-worth ($25bn) of assets and cash change hands.
July 10, 2016 Comments Off on Sanofi has agreed to exchange its Merial animal health business for Boehringer Ingelheim’s consumer healthcare unit in a deal that will see €21.8bn-worth ($25bn) of assets and cash change hands.
The deal ties in with the current trend among pharma companies to rationalise their businesses and focus on fewer key product categories. Sanofi will transfer ownership of the €11.4bn Merial division to Boehringer in return for the latter’s consumer health unit – valued at €6.7bn – and €4.7bn in cash. Contracts to start the process have been signed and the transaction is expected to close before the end of the year, according to the two companies, which have been negotiating the asset swap deal since December 2015. Both the animal and consumer health sectors have seen considerable consolidation in the last few years, as companies strive to boost profitability by broadening product ranges, reducing duplicate costs and expanding in overseas markets. The veterinary sector saw Pfizer spin out its Zoetis unit – the biggest player in the market – in 2011, while Elanco bought Novartis’ unit last year to claim the second spot ahead of Merck/MSD Animal Health. It is estimated that combining Merial with Boehringer’s smaller operations will allow the combined business to leapfrog into the second-place spot in terms of market share, with pro forma sales of around €3.8bn in 2015. In consumer health, the deal comes in…
Continue reading …»السعودية – ولي ولي العهد يسلم أول ترخيص استثمار أجنبي 100% لـ«داو كميكال
June 19, 2016 Comments Off on »السعودية – ولي ولي العهد يسلم أول ترخيص استثمار أجنبي 100% لـ«داو كميكال
سلم ولي ولي العهد النائب الثاني لرئيس مجلس الوزراء وزير الدفاع رئيس مجلس الشؤون الاقتصادية والتنمية الأمير محمد بن سلمان، أول ترخيص استثماري في القطاع التجاري بملكية أجنبية 100 في المئة، إلى رئيس شركة «داو كميكال» الأميركية أندرو ليفيريس، خلال استقباله في واشنطن أمس، في حضور كل من وزير التجارة والاستثمار رئيس مجلس إدارة الهيئة العامة للاستثمار الدكتور ماجد القصبي، ووزير الطاقة والصناعة والثروة المعدنية المهندس خالد الفالح، وذلك بعد أيام من موافقة مجلس الوزراء على فتح مجال الاستثمار في نشاط تجارة الجملة والتجزئة لجميع الشركات الأجنبية العاملة في هذا القطاع. وتعدّ شركة «داو كميكال»، المدرجة في مؤشر داوجونز الأميركي وأسست قبل نحو 120 عاماً، من كبرى الشركات العالمية في مجال الصناعات البتروكيماوية والصناعات التحويلية وفي مجال الابتكار والبحوث، وحققت إيرادات سنوية في 2015 حوالى 48 بليون دولار، ويبلغ عدد موظفيها 51 ألف موظف. وأكدت الهيئة العامة للاستثمار، في بيان لها أمس، (حصلت «الحياة» على نسخة منه) أن وجود شركة «داو كميكال» في المملكة سيسهم بشكل كبير في إضافة مردود عالٍ ونوعي إلى قطاع الصناعات البتروكيماوية»، موضحة أن «الشروط اللازمة للترخيص للشركات الأجنبية للاستثمار في قطاع تجارة الجملة والتجزئة بنسبة ملكية 100 في المئة نصت على الترخيص للشركات التي لديها وجود في ثلاث أسواق في حد أدنى، وألّا يقل رأس…
Continue reading …Biogen, AbbVie once-monthly MS injection wins U.S. FDA approval
May 29, 2016 Comments Off on Biogen, AbbVie once-monthly MS injection wins U.S. FDA approval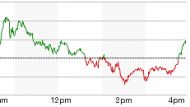
The U.S. Food and Drug Administration on Friday approved a once-monthly injection for multiple sclerosis from Biogen Inc and AbbVie Inc. The self-administered, under-the-skin injection, Zinbryta, is designed to treat adults with relapsing forms of the disease. source : Reuters 29 – May – 2016
Continue reading …RANKED: These are the most and least reputable drug companies in the world
May 26, 2016 Comments Off on RANKED: These are the most and least reputable drug companies in the world
Drug companies aren’t exactly known for their reputations. But public opinions about them have changed in some surprising ways in recent years, especially amidst recent controversies over the high and rising prices of drugs. A new analysis from the research consulting firm Reputation Institute evaluated public perceptions of 14 pharmaceutical companies in 7 key areas: 1) products and services, 2) innovation, 3) workplace, 4) governance, 5) citizenship, 6) leadership, and 7) financial performance. The report found that on average, drug companies continue to be perceived as having a so-so reputation. In fact, people’s opinion of the pharmaceutical industry overall has improved since 2015. Yet Kasper Ulf Nielsen, an executive partner at the Reputation Institute, told Business Insider that these average ratings hide some of the more interesting aspects of the results. For one thing, people tend to have polar opposite views of individual companies, either ranking them as having an “excellent” or “poor” reputation. The company with the highest reputation was Bayer. Also, young people aged 18-24 had a significantly lower impression of pharmaceutical companies than did older people. Check out the Reputation Institute’s drug company scores, ranked from lowest to highest: Pfizer. RepTrak Points: 65.9 Pfizer, which is known for drugs like Lipitor and Viagra, was ranked the second-most familiar of the 14 drug companies analyzed by the…
Continue reading …Novartis splits drugs business into two, pharma chief to leave
May 18, 2016 Comments Off on Novartis splits drugs business into two, pharma chief to leave
Novartis is splitting its pharmaceuticals division into two business units, one focused on cancer and the second on other drugs, while switching out its current pharma head in the second high-profile management reshuffle this year. David Epstein, the American head of Novartis Pharmaceuticals and a 27-year veteran with the group, will leave the company to “explore new challenges from the U.S.”, Novartis said. Epstein’s re-location to the United States implies he is not in the running to replace Andrew Witty as chief executive of British drugmaker GlaxoSmithKline next year, as some have speculated. GSK has said it expects to choose a new CEO towards the end of the 2016. Novartis’ reorganization of its main drugs unit, which accounts for about two-thirds of its $49 billion in annual sales, shows the growing importance of oncology to the company, after it bought GSK’s marketed cancer drugs for $16 billion last year. It comes as Novartis struggles with the patent expiration of blood cancer drug Glivec and slower-than-expected revenue from its new heart failure medicine, Entresto. Epstein is the second high-ranking Novartis official to exit within months. Ex-Hospira chief Michael Ball replaced Jeff George at the company’s struggling Alcon eye care business in…
Continue reading …Pfizer to buy Anacor for around $5.2 billion
May 17, 2016 Comments Off on Pfizer to buy Anacor for around $5.2 billion
Pfizer entered a definitive merger agreement to acquire Anacor Pharmaceuticals for $99.25 per share in cash, or a total transaction value of approximately $5.2 billion, the companies announced Monday. The deal, which represents a premium of about 55 percent to Anacor’s closing share price on May 13 and has been approved by the board of directors of both drugmakers, gives Pfizer rights to the non-steroidal topical PDE4 inhibitor crisaborole. Albert Bourla, group president of Pfizer’s Global Innovative Pharma and Global Vaccines, Oncology and Consumer Healthcare businesses, said “crisaborole is a differentiated asset with compelling clinical data that, if approved, has the potential to be an important first-line treatment option.” A marketing application for crisaborole is currently under review by the FDA for the treatment of mild-to-moderate atopic dermatitis, with a target review date of January 7, 2017. In study data unveiled last year, crisaborole was shown to provide significantly greater skin clearance than placebo. “Anacor will be a strong fit with Pfizer’s innovative business, further supporting our strategic focus on inflammation and immunology,” Bourla remarked, adding “we believe we are well positioned to maximize crisaborole’s commercial potential through our strong relationships with paediatricians and primary care physicians.” Pfizer suggested that if…
Continue reading …AstraZeneca heart drug fails in key stroke trial
May 12, 2016 Comments Off on AstraZeneca heart drug fails in key stroke trial
AstraZeneca’s heart drug Brilinta has failed to help stroke patients as hoped in a big clinical trial, dealing a blow to the company’s expectations that the medicine could rack up annual sales of $3.5 billion by 2023. The trial found the blood-thinner was not significantly better than aspirin, the current standard of care, in preventing recurrent attacks in the 90 days after patients suffer a stroke, the British drugmaker said on Wednesday. AstraZeneca had hoped the study would open up a new market for Brilinta beyond its current use in treating heart attack patients. Several analysts had thought there was a good chance of success, since Sanofi’s now off-patent drug Plavix previously showed limited benefit in strokes and Brilinta is more potent. In the event, fewer patients taking AstraZeneca’s drug in the trial had a recurrent stroke, suffered a heart attack or died than those on aspirin, but the difference was not statistically significant. A positive result would have boosted consensus forecasts for Brilinta, which currently stand at an annual $1.87 billion for 2020, according to Thomson Reuters Cortellis. AstraZeneca’s own projection of $3.5 billion by 2023 makes Brilinta an important part of a $45 billion revenue target announced by…
Continue reading …Pharmaceutical Giant Novartis Facing Lawsuit Over Bribing Doctors to Prescribe Their Drugs
April 11, 2016 Comments Off on Pharmaceutical Giant Novartis Facing Lawsuit Over Bribing Doctors to Prescribe Their Drugs
Novartis AG and the Southern District of New York — which oversees Wall Street — are currently engaged in a whistleblower lawsuit. The U.S. alleges the Swiss pharmaceutical company has been wining and dining doctors at phony speaking events as a form of kickback for over a decade. Last year, Novartis settled a separate suit with the U.S. In that case, filed in 2013, the government accused the company of sending patients to “specialty pharmacies” who, in exchange for rebates, would recommend Novartis drugs to customers. Then, claimed the government, these specialty pharmacies submitted thousands of what Bloomberg called “fraud-tainted reimbursement claims” to Medicare and Medicaid — to the tune of half a billion dollars. “Novartis corrupted the prescription drug dispensing process,” said Preet Bharara, U.S. Attorney for Manhattan, in a statement back in 2013. “For its investment, Novartis reaped dramatically increased profits on these drugs, and Medicare, Medicaid and other federal healthcare programs were left holding the bag.” Between the individual fines on each “fraud-tainted” claim and the state seeking triple damages, the U.S. originally sought $3.3 billion from Novartis. That case settled in November of 2015, with the corporation agreeing to pay $390 million. But with Manhattan now asking for files on 80,000…
Continue reading …“البورصة” تنشر مبيعات أكبر 20 شركة دواء فى ٢٠١٥
March 10, 2016 Comments Off on “البورصة” تنشر مبيعات أكبر 20 شركة دواء فى ٢٠١٥
31.7 مليار جنيه حجم السوق بزيادة %13.. والنمو بالدولار يدخل مرحلة الخطر “الحكمة” تطيح بـ”جلوبال نابى” و”MSD” من قائمة الشركات الـ10 الأعلى مبيعاً.. وسيجما تختفى حققت شركات الادوية العاملة بالسوق المصرى، مبيعات إجمالية بقيمة 4.1 مليار دولار (31.7 مليار جنيه)، خلال الفترة من 1 يناير إلى 31 ديسمبر 2015، بنمو %13 بالجنيه المصرى، و %4 بالدولار. وكشف تقرير صادر عن مؤسسة “IMS” العالمية للمعلومات والاستشارات فى مجال الصيدلة والرعاية الطبية، حصلت “البورصة” على نسخة منه، عن استحواذ 20 شركة أدوية على أكثر من %55 من مبيعات السوق العام الماضى. وتضم قائمة الـ20 شركة الأكثر مبيعاً، 11 شركة أجنبية، تستحوذ على نحو ثلث مبيعات السوق (11.8 مليار جنيه)، مقابل 9 شركات محلية تبلغ مبيعاتها 6 مليارات جنيه. وأوضح التقرير، أن 10 شركات أدوية فقط هى (نوفارتس، جلاكسو، سانوفى، فاركو، إيبيكو، أمون، فايزر، ايفا، ماركيرل، الحكمة) تسيطر على %38.7 من السوق، بحجم مبيعات بلغ 12.2 مليار جنيه، فيما حققت الشركات الـ10 الأخرى بالقائمة وهى (جلوبال نابى، MSD، أبوت، مالتى إيبيكس، ميرك، استرازينكا، سيرفى، المهن الطبية، باير، سيديكو)، مبيعات بقيمة 5.5 مليار جنيه تعادل %17 من السوق. وعلى الرغم من حفاظ السوق على نسب نموه الطبيعية التى تتراوح بين 12 و%15 بالجنيه المصرى، لكن النمو بالدولار دخل مرحلة الخطر، بحسب مصادر ببعض الشركات…
Continue reading …FDA gives ocrelizumab breakthrough designation for PPMS: Roche
February 18, 2016 Comments Off on FDA gives ocrelizumab breakthrough designation for PPMS: Roche
The U.S. Food and Drug Administration has granted breakthrough therapy designation for ocrelizumab (OCREVUS) for treating people with primary progressive multiple sclerosis (PPMS), Swiss drugmaker Roche said on Wednesday. There are no approved treatments now for PPMS, a debilitating form of the disease marked by steadily worsening symptoms. “Roche plans to pursue marketing authorization for both PPMS and relapsing multiple sclerosis (RMS), a more common form of the disease, and will submit data from three pivotal Phase III studies to global regulatory authorities in the first half of 2016,” it said in a statement. Roche believes ocrelizumab’s potency and benign side effect profile make it unique, while industry analysts see it as a likely multibillion-dollar-a-year seller that will help the group diversify beyond its mainstay cancer business. ADVERTISEMENT Breakthrough therapy designation expedites the development and review of medicines intended to treat serious or life-threatening diseases. Ocrelizumab is the first investigational medicine for MS to win the designation by the FDA, Roche said. Clinical trials released in October showed Roche’s big new drug hope cut multiple sclerosis relapses by nearly half compared with the older product Rebif, underscoring its potential in the main relapsing form of the disease. Ocrelizumab also cut…
Continue reading …Pfizer to create new division following Allergan acquisition
February 10, 2016 Comments Off on Pfizer to create new division following Allergan acquisition
Pfizer on Monday announced that following the completion of its $160-billion acquisition of Allergan later this year, a new operating segment named global specialty and consumer brands will be created, led by Bill Meury, current president of Allergan’s branded pharmaceutical unit. Pfizer indicated that the new division will include its consumer healthcare unit and Allergan’s ophthalmology and aesthetics businesses, and Botox therapeutic and cosmetic. The drugmaker also revealed that after the completion of the transaction, its vaccines and oncology businesses will be combined with its global innovative pharmaceutical division, with the combined unit headed by Albert Bourla, who currently leads Pfizer’s vaccines, oncology and consumer segment. Bourla will report directly to current Allergan CEO Brent Saunders, who, as previously disclosed, will become Pfizer’s president and chief operating officer at the close of the deal. Pfizer added that following the close of the transaction, its commercial operations will continue through two distinct businesses, namely an innovative products business and an established products business. The innovative products business will consist of the global innovative pharmaceutical and global specialty and consumer brands segments, while the established products business, which will be headed by John Young, will include the global established pharmaceutical segment, including…
Continue reading …U.S. patent office rules against Amgen Humira challenge
January 18, 2016 Comments Off on U.S. patent office rules against Amgen Humira challenge
U.S. patent officials on Thursday denied petitions by Amgen to review two formulation patents on AbbVie’s Humira, a potential setback in Amgen efforts to market a biosimilar version of the world’s top-selling prescription medicine. In June, Amgen, the world’s biggest biotechnology company, asked the U.S. Patent and Trademark Office for the review, arguing that the patents in question should not have been granted in the first place for Humira, an injected rheumatoid arthritis treatment with annual sales approaching $14 billion. In declining to review the patents, the agency said “we determine, based on the petition and the accompanying evidence, that Amgen has not shown a reasonable likelihood of prevailing on any of its challenges.” Amgen said it still plans to challenge the legality of the patents. “Although we are disappointed by the PTO’s decision, we continue to question the validity of the patents,” the company said in a statement. “We look forward to presenting our position in court.” Source : Reuters 18 – January – 2016
Continue reading …Pfizer Hikes Prices for Over 100 Drugs on January 1
January 10, 2016 Comments Off on Pfizer Hikes Prices for Over 100 Drugs on January 1
Pfizer Inc, which plans a $160-billion merger with Ireland-based Allergan Plc to slash its U.S. tax bill, on Jan. 1 raised U.S. prices for more than 100 of its drugs, some by as much as 20 percent, according to statistics compiled by global information services company Wolters Kluwer. Pfizer confirmed a 9.4 percent increase for heavily advertised pain drug Lyrica, which generated $2.3 billion in 2014 U.S. sales; a 12.9 percent increase for erectile dysfunction drug Viagra, which had 2014 U.S. sales of $1.1 billion; and a 5 percent increase for Ibrance, a novel breast cancer drug launched last year at a list price of $9,850 per month, or $118,200 per year. Company spokesman Steven Danehy could not immediately confirm the remaining price increases, which were compiled by a unit of Wolters Kluwer Health and published in a research note by UBS Securities. U.S. lawmakers, and presidential candidates, have in recent months stepped up criticism of U.S. drug prices trends, driven in part by eye-popping price hikes from companies with recently acquired generic drugs. “Medicines are among the most effective and efficient use of private and public health care dollars,” Pfizer said in an emailed statement. “It is important to…
Continue reading …Johnson & Johnson’s Stelara succeeds in Phase III Crohn’s disease trial
December 26, 2015 Comments Off on Johnson & Johnson’s Stelara succeeds in Phase III Crohn’s disease trial
Johnson & Johnson’s Stelara was significantly better than placebo at inducing clinical response and remissions in patients with moderate to severe Crohn’s disease, according to data from a late stage trial, providing ammunition for a potential expanded approval of the medicine. Stelara, a biotech medicine that blocks inflammation, is approved to treat the skin condition scaly plaque psoriasis and a type of arthritis associated with psoriasis. J&J reported $613 million in third-quarter Stelara sales. The company said it was on track for global approval filings for Crohn’s this year. Subjects in the 628-patient trial presented on Monday received either a single infusion of 130 milligrams of Stelara, a Stelara infusion of 6 mg per kilogram of weight or a placebo. After six weeks, 52 percent of those in the 130 mg group and 56 percent in the 6mg/kg group experienced a clinical response, defined as a reduction from baseline of at least 100 points in the Crohn’s Disease Activity Index score. That compared with 29 percent in the placebo group. In addition, 31 percent of the Stelara 130 mg group and 40 percent in the other dosing arm achieved clinical remission by week 8 of the study, versus 20 percent…
Continue reading …Sanofi plans to reshape via $20 billion asset swap with Boehringer
December 17, 2015 Comments Off on Sanofi plans to reshape via $20 billion asset swap with Boehringer
Sanofi and Boehringer Ingelheim are in exclusive talks over a $20 billion swap of the French pharmaceuticals company’s animal health business for the family-owned German group’s consumer health operation. The deal would consist of an exchange of Sanofi’s Merial animal health arm, with an enterprise value of 11.4 billion euros ($12.6 billion), with Boehringer’s consumer health division, worth 6.7 billion. Boehringer would also pay 4.7 billion euros in cash to Sanofi, the companies said on Tuesday. The plan signals a radical reshaping of Sanofi under new boss Olivier Brandicourt, who took over in April, and has said he sees “limited synergies” between animal health and the rest of the business. The deal would vault the French drugmaker into number one spot in the fragmented consumer healthcare (CHC) marketplace, with proforma 2015 sales of approximately 5.1 billion euros and a global market share of around 4.6 percent. Boehringer would become the world’s second-largest animal health company. The global pharmaceutical industry has seen a flurry of deal-making in the past two years, as large companies try to focus on a smaller number of businesses where they can establish a leading position. “In entering into exclusive negotiations with Boehringer Ingelheim, we have acted…
Continue reading …AstraZeneca boosts respiratory unit with $575 million Takeda deal
December 17, 2015 Comments Off on AstraZeneca boosts respiratory unit with $575 million Takeda deal
Britain’s AstraZeneca (AZN.L) has agreed to buy Takeda Pharmaceutical’s (4502.T) respiratory business, including expanded rights to roflumilast, a treatment for chronic obstructive pulmonary disease (COPD). AstraZeneca, which has marketed roflumilast in the United States under the name Daliresp since the first quarter of 2015, said it would pay $575 million to the Japanese company. “Full acquisition of the global rights will support AstraZeneca’s respiratory franchise and complement the company’s portfolio of treatments for severe COPD,” AstraZeneca said. Roflumilast, known as Daxas outside the US, is the only oral PDE4 inhibitor, a type of drug that has anti-inflammatory effects, approved for the treatment of COPD. AstraZeneca acquired the US rights for the drug from Actavis earlier this year as part of a deal that bolstered its respiratory business, one of the company’s three main areas of focus. Annual global sales of the three core medicines acquired — Alvesco, Omnaris and Daxas — were $198 million for the period ending in March 2015, AstraZeneca said. Earlier on Wednesday, AstraZeneca said it would spend $800 million to expand its presence in China over the next 10 years. Rothschild acted for Takeda on the deal. 17 – December – 2015
Continue reading …AstraZeneca in advanced talks to buy Acerta Pharma
December 14, 2015 Comments Off on AstraZeneca in advanced talks to buy Acerta Pharma
AstraZeneca confirmed Monday that it is “exploring potential strategic options” with Acerta Pharma following recent speculation. AstraZeneca said “there can be no certainty that any transaction will ultimately be entered into,” adding that it “will make a further announcement if and when appropriate.” According to people with knowledge of the situation, AstraZeneca is in advanced negotiations to acquire cancer-drug developer Acerta for more than $5 billion, The Wall Street Journal reported Friday. Some of the sources said the companies hope to reach an agreement by the end of the month, although they cautioned there is no assurance a deal will be reached. Acerta’s lead compound acalabrutinib, also known as ACP-196, is currently under development for the treatment of various malignancies, including leukaemias and lymphomas. Earlier this month, the company announced Phase I/II data showing that the Bruton tyrosine kinase (BTK) inhibitor was associated with a response rate of 95 percent among patients with relapsed chronic lymphocytic leukaemia. The experimental drug is also being investigated in combination with Merck & Co.’s immunotherapy Keytruda (pembrolizumab). Acalabrutinib works in a manner similar to AbbVie and Johnson & Johnson’s Imbruvica (ibrutinib), which was a key asset in the company’s move to purchasePharmacyclics for $21 billion earlier this year. Acerta…
Continue reading …سبعة عشر شركة جديدة تطلب إنتاج سوفالدى المصرى
December 3, 2015 Comments Off on سبعة عشر شركة جديدة تطلب إنتاج سوفالدى المصرى
ذكر الدكتور طارق سالمان، مساعد وزير الصحة لشؤون الدواء، إن 17 شركة أدوية قدمت طلبات للحصول على تصريح بإنتاج عقار «سوفالدى» المصرى، المخصص لعلاج مرضى الالتهاب الكبدى الوبائى «سى»، مشيراً إلى أنه سيتم السماح لها فور التأكد من توافر اشتراطات الجودة ومعايير الأمان والفاعلية، ليرتفع العدد إلى 32 شركة، بينها 13 شركة بدأت ضخ منتجاتها في السوق بالفعل، و2 في المراحل الأخيرة للإنتاج. وأضاف، أن الوزارة وضعت العديد من الإجراءات الصارمة لضمان جودة وفاعلية العقار، تبدأ بالتأكد من التصنيع الجيد، ومطابقة الاشتراطات للمادة الخام التي تستوردها الشركة، وتحليل المادة بهيئة الرقابة الدوائية، ثم تحليل عينة عشوائية من المنتج النهائى، مؤكدا أنه لا يُسمح للشركة بالإفراج عن أول 3 تشغيلات للمنتج إلا بعد التأكد من سلامة نتائج التحليل. وأجاب «سالمان» عن سؤال عن السماح لعدد كبير من الشركات بالإنتاج بقوله إن الأصل هو تحديد 10 شركات للعقار الواحد، لكن الوزير الدكتور أحمد عماد سمح بزيادة العدد لتلبية احتياجات السوق، وعلاج أكبر عدد ممكن من المرضى بالأدوية المحلية التي ثبتت كفاءتها وفاعليتها مقارنة بالدواء المستورد، موضحاً أن سعر العقار في الصيدليات موحد، وهو 1600 جنيه، متوقعاً تخفيضه في المناقصة القادمة. في سياق متصل، قدم الدكتور محيى عبيد، نقيب الصيادلة، مذكرة لوزير الصحة يطالبه فيها بالتحقيق في تصريحات الدكتور يحيى الشاذلى، مستشار الوزير…
Continue reading …Trump on Pfizer-Allergan: ‘Our Politicians Should Be Ashamed’
November 26, 2015 Comments Off on Trump on Pfizer-Allergan: ‘Our Politicians Should Be Ashamed’
“The fact that Pfizer is leaving our country with a tremendous loss of jobs is disgusting. Our politicians should be ashamed,” Republican presidential polling frontrunner Donald Trump says in a statement. 26 – November – 2015
Continue reading …أكبر عشر شركات أدوية في العالم تحقق إيرادات ب 444 مليار دولار
November 26, 2015 Comments Off on أكبر عشر شركات أدوية في العالم تحقق إيرادات ب 444 مليار دولار
26 – November – 2015
Continue reading …Exclusive: Pfizer, Allergan CEOs agree on combined company roles – sources
November 24, 2015 Comments Off on Exclusive: Pfizer, Allergan CEOs agree on combined company roles – sources
Pfizer Inc and Allergan Inc chief executives have agreed on the roles they would assume in a combined company, removing one of the last hurdles to the largest ever healthcare merger, people familiar with the matter said. Pfizer CEO Ian Read will be CEO of the combined company, while Allergan CEO Brent Saunders will have another very senior role, the people said on Friday, without providing more details. A deal announcement is expected in the coming days, one of the people added. The sources asked not to be identified because the negotiations are confidential. Pfizer and Allergan declined to comment. A top job at the combined company would position Saunders, 45, to have a claim at the CEO job when Read, 62, steps down. This arrangement also shows that Pfizer’s management plans to remain in control, at least in the short term, even if the deal is technically structured so that Allergan buys Pfizer. The roughly $150 billion merger would see New York-based Pfizer redomicile in Ireland, where Botox-maker Allergan is registered. The U.S. Treasury Department on Thursday took steps to clamp down on tax-avoiding inversion deals with new rules, though there was scarce evidence it would stop the biggest…
Continue reading …فايزر تشتري “أليرغان” في أكبر صفقة في تاريخ صناعة الأدوية
November 24, 2015 Comments Off on فايزر تشتري “أليرغان” في أكبر صفقة في تاريخ صناعة الأدوية
وافقت شركة فايزر الأمريكية العملاقة للأدوية على صفقة لشراء شركة اليرغان مقابل 160 مليار دولار، لتكون بذلك أكبر صفقة في تاريخ صناعة الأدوية. وسيؤدي هذا الاندماج إلى تأسيس أكبر شركة لإنتاج الأدوية في العالم. وكانت الشركتان قد تفاوضتا في هذا الشأن على مدى الأسابيع الماضية. وتوقع محللون أن تسمح هذه الصفقة لشركة فايزر بتفادي رسوم الضرائب الباهظة نسبيا في الولايات المتحدة من خلال نقل مقرها إلى دبلن. وكانت فايزر قد تقدمت بعرض لشراء شركة استرازينيكا البريطانية لصناعة الأدوية، لكن الأخيرة رفضت الطلب، مشيرة إلى أن العرض أقل من قيمة الشركة الحقيقية، وأفقها المستقبلي. وسيحصل المساهمون في اليرغان على 11.3 من أسهم فايزر لكل سهم من أسهمهم في اليرغان. وسيدير الشركة الجديدة المدمجة الرئيس التنفيذي لفايزر، إيان ريد، بينما سيتولى برنت ساندرز رئيس اليرغان منصب النائب. وستكون هذه الصفقة هي الأحدث في سلسلة من عمليات الدمج والاستحواذ في قطاع الأدوية في الوقت الذي تسعى فيه شركات الأدوية لمعالجة مشكلة نفاد صلاحية براءات الاختراع لعدد من الأدوية الرئيسية، في حين أدى التحسن في فهم علوم الوراثة والأمراض إلى إنتاج المزيد من العقاقير الموجهة التي لها سوق أصغر حجما. بالعربى BBC المصدر 24 – November – 2015
Continue reading …تشغيل مصنع “ميدل إيست” للأدوية العام المقبل باستثمارات 50 مليون جنيه
November 16, 2015 Comments Off on تشغيل مصنع “ميدل إيست” للأدوية العام المقبل باستثمارات 50 مليون جنيه
خمسة وعشرون مليون جنيه مبيعات مستهدفة للشركة بعد تشغيل المصنع.. وإنتاج أدوية بشرية وبيطرية ومكملات غذائية مليارا جنيه حجم استثمارات 1200 شركة تصنع لدى الغير.. و30% من الشركات معروضة للبيع غنيم: بنود اتفاقية سحب الأدوية المنتهية “كوارث” والموقعون عليها ارتكبوا جريمة عودة تراخيص الشركات التجارية مرة أخرى نهاية العام الحالى بشروط أصعب ارجأت شركة “ميدل إيست” للأدوية، تشغيل مصنعها الجديد بمدينة العبور للربع الثالث من العام المقبل، لعدم انتهاء الإنشاءات، واستيراد خطوط الإنتاج، نتيجة ارتفاع تكاليف الإنتاج بسبب ازمة الدولار فى الفترة الاخيرة. وقال محمد غنيم، رئيس مجلس ادارة الشركة، ورئيس النقابة العامة لمصنعى المستحضرات الدوائية لدى الغير”التول”، إن تكرار ارتفاع سعر الدولار مقابل الجنيه منذ بداية العام، رفع تكلفة الإنشاءات وخطوط الإنتاج بشكل كبير، ما أجبر الشركة على تأجيل تشغيل المصنع للمرة الثانية. ويقع المصنع الجديد على مساحة 5200 متر، ويضم 7 خطوط لإنتاج المراهم والكريمات والأقراص والكبسولات الصلبة، وتقدر استثماراته المبدئية بنحو 50 مليون جنيه، وفقاً لغنيم. وبدأت “Middle east” إنشاءات مصنعها بالعبور عام 2009، وتوقفت مطلع 2011 بسب أحداث ثورة 25 يناير، وعاودت الإنشاءات مرة أخرى 2012، لكن الظروف الاقتصادية حالت دون التشغيل الفعلى للمصنع. ويستهدف المصنع إنتاج أدوية بشرية وبيطرية ومستحضرات عناية شخصية ومكملات غذائية، وانتهت الشركة من تسجيل 15 عقاراً بإدارة الصيدلة بوزارة الصحة، فيما تمتلك…
Continue reading …GlaxoSmithKline targets filing 20 new drugs for approval by 2020
November 6, 2015 Comments Off on GlaxoSmithKline targets filing 20 new drugs for approval by 2020
GlaxoSmithKline said Tuesday at an investor presentation that it hopes to file up to 20 new drugs with regulators before 2020, including seven medicines that are currently in late-stage development and could be launched before the end of the decade. CEO Andrew Witty commented “today, we have profiled around 40 innovative potential new medicines and vaccines which will support future growth in our pharmaceuticals and vaccines businesses.” He added “for the first time, we have also outlined the scale of new opportunities…in earlier stages of development, notably in areas such as oncology and immuno-inflammation.” GlaxoSmithKline noted that it can potentially initiate Phase II studies for approximately 30 new chemical entities and product line extensions by 2017 and start Phase III development of up to 20 new chemical entities and product line extensions. The company, which is focused on the core areas of HIV and other infections, respiratory medicine, oncology, immuno-inflammation, vaccines and rare diseases, added that it additionally plans to begin late-stage studies of up to 20 assets between 2021 and 2025. “The level of innovation in this portfolio is substantial,” Witty stated, adding “we believe this is critical in today’s operating environment as payers look to balance pressures of…
Continue reading …3 Drugs That Are About to Become Billion-Dollar Blockbusters in 2016
November 6, 2015 Comments Off on 3 Drugs That Are About to Become Billion-Dollar Blockbusters in 2016
Bristol-Myers Squibb, Pfizer, and AbbVie Inc. market some of the fastest-growing cancer drugs on the market. It’s the hope of every drug developer to create a billion-dollar-per-year blockbuster therapy, but with 90% of drugs failing in clinical trials, few drug companies successfully do it. Bristol-Myers Squibb (NYSE:BMY), Pfizer (NYSE:PFE), and AbbVie (NYSE:ABBV), however, appear to have beaten those long odds, because each has a drug that’s on pace to eclipse blockbuster status in the coming year. Bristol-Myers Squibb: Opdivo Bristol-Myers’ immuno-oncology drug Opdivo, which costs about $12,500 per month, trounced industry watchers’ estimates in the third quarter. Revenue from Opdivo clocked in at $305 million, and that was nicely higher than the $238 million that had been expected. Opdivo’s sales success stems from a massive R&D effort from Bristol-Myers to establish it as a go-to anti-cancer therapy that can be used both alone and in combination with other widely used drugs to make them work better. So far, Opdivo has been approved for use as a monotherapy in the treatment of advanced melanoma and lung cancer and alongside another Bristol-Myers drug, Yervoy, in a specific genetic variation of metastatic melanoma. More approvals are likely, too, because Bristol-Myers is conducting more…
Continue reading …أكبر صفقات 2015 ستكون بقطاع التجميل
November 2, 2015 Comments Off on أكبر صفقات 2015 ستكون بقطاع التجميل
قالت صحيفتا وول ستريت جورنال وفايننشال تايمز إن شركة فايزر العملاقة للأدوية تجري محادثات في مراحلها المبكرة مع شركة أليرجان المصنعة للبوتوكس، لبحث ما قد تصبح أكبر صفقة استحواذ هذا العام. وإذا تقدمت فايزر بعرض لشراء أليرجان التي تبلغ قيمتها السوقية 113 مليار دولار فستكون هذه هي ثاني محاولة تقوم بها فايزر في الآونة الأخيرة للاستحواذ على منافس كبير لها بعد محاولتها التي باءت بالفشل لشراء مجموعة أسترا زينيكا البريطانية السويدية للأدوية في العام الماضي. ومن شأن اندماج أليرجان وفايزر التي تبلغ قيمتها 219 مليار دولار أن يتمخض عن أكبر مجموعة للرعاية الصحية في العالم تبلغ قيمتها السوقية نحو 330 مليار دولار لتتفوق على جونسون آند جونسون البالغة قيمتها 278 مليار دولار. وقال متحدث باسم فايزر أكبر شركة لصناعة الأدوية في الولايات المتحدة إنه لا يعلق على “الشائعات والتكهنات في السوق”. وأحجمت أليرجان أيضا عن التعقيب. وإذا تمت الصفقة فإن أليرجان ستعزز نمو إجمالي إيرادات فايزر المتوقع أن تتراجع إيراداتها 3.3 بالمئة هذا العام. ومن المتوقع أن ترتفع إيرادات أليرجان 39 بالمئة هذا العام، وفقا لتقديرات تومسون رويترز آي/بي/إي/إس. وقالت صحيفة وول ستريت جورنال إن محادثات الاندماج في مراحلها المبكرة وقد لا تثمر عن اتفاق، بينما لم تتضح أي تفاصيل أخرى. ونشرت صحيفة فايننشتال تايمز نبأ المحادثات في وقت لاحق…
Continue reading …إضراب العاملين بمصنع جلاكسو للأدوية بعد تسريح 110 عامل
October 29, 2015 Comments Off on إضراب العاملين بمصنع جلاكسو للأدوية بعد تسريح 110 عامل
أعلن المجلس المصرى لحقوق العمال والفلاحين عن تضامنه مع إضراب أكثر من 700 عامل بمصنع شركة جلاكسو سيمثكلاين للأدوية بمدينة السلام بالقاهرة، بعد تسريح 110 من العاملين الشهر الماضى بدون منحهم مستحقاتهم المالية عن فترات خدمتهم. أكد حسام فودة، رئيس المجلس المصرى، فى تصريحات خاصة لـ”اليوم السابع”، أن العاملين لا تطبق عليهم الزيادة السنوية للرواتب بنسبة 10% كما يقر القانون لكن الزيادة 3% فقط، لافتًا إلى أنه لا يوجد نظام محدد لمكافأة نهاية الخدمة للعاملين ما يدفع الشركة لمنح صغار العاملين مستحقات مالية متفاوتة بدون قواعد قانونية وفقا لما هو محدد بقانون العمل رقم 12 لسنة 2003 وتعديلاته. وقال فودة، إن العاملين ليس لديهم فى المصنع تنظيم نقابى يدافع عنهم على الرغم من أن زملاءهم فى فرع الهرم بالجيزة لديهم تنظيم نقابى لذلك تقل الاحتجاجات بسبب المطالَب المشروعة. وأوضح أنه فى الوقت الذى منعت عن العاملين امتيازات مالية مثل منح الأعياد والإجازات الرسمية يومى الجمعة والسبت إلا أن الإدارة قامت بشراء سيارات فارهة لمديرى الإدارات العليا. واستنكر تهديد العاملين من قبل إدارة مصنع السلام باستدعاء قوات الأمن لفض احتجاجهم السلمى، خاصة بعد ان رفض العاملون التفاوض بشكل فرادى خوفا من التنكيل بهم وتقديم كبش فداء للإدارة كما حدث من قبل. المصدر : جريدة اليوم السابع 29 – October – 2015
Continue reading …Doomed cholesterol drug causes Eli Lilly’s worst day since 2008
October 15, 2015 Comments Off on Doomed cholesterol drug causes Eli Lilly’s worst day since 2008
Eli Lilly just suffered its darkest day since the 2008 financial crisis. The drug maker’s shares plunged 8% on Monday after it scrapped a potential blockbuster experimental cholesterol drug. Eli Lilly (LLY) said the drug, known as evacetrapib, is being abandoned because research showed the medicine has failed to treat high-risk heart disease. Late-stage drug trials in about 12,000 patients showed “insufficient efficacy,” Lilly said. “We’re obviously disappointed in this outcome, as we hoped that evacetrapib would offer an advance in treatment for people with high-risk cardiovascular disease,” David Ricks, president of Lilly Bio-Medicines, said in a statement. $6 billion in Lilly market cap vanishes It’s a big setback and has spooked investors. The stock selloff wiped out roughly $6 billion from Lilly’s market value. The stock suffered its worst one-day percentage decline since December 2008. Previous research raised hopes on Wall Street and in the medical community that evacetrapib could be a big success in preventing heart attacks, strokes and other cardiovascular problems. A study presented at the American Heart Association’s annual meeting in late 2011 showed the treatment — part of a class of drugs known as CETP inhibitors — was able to enhance good cholesterol levels and…
Continue reading …رئيس مجلس إدارة «نوفو نورديسك» الدنماركية : طرح الجيل الثانى من الأنسولين فى مصر عام 2016
September 14, 2015 Comments Off on رئيس مجلس إدارة «نوفو نورديسك» الدنماركية : طرح الجيل الثانى من الأنسولين فى مصر عام 2016
• لارس سورينسن: استثماراتنا الجديدة بمصر تحقق لها الاكتفاء الذاتى من دواء مرض السكر • «نوفو كير» برنامج الشركة المجانى لتوعية المصريين بكيفية التعامل مع المرض أكثر من مليون ونصف المليون مريض بالسكر يستخدمون الأنسولين فى مصر، و70% منهم يحصلون عليه من شركة «نوفو نورديسك» الدنماركية، التى يمتد تواجدها فى مصر لأكثر من 80 عاما، فهى أول شركة تقوم بإدخال الأنسولين إلى مصر عام 1933، بحسب لارس ريبين سورينسن رئيس مجلس الإدارة والرئيس التنفيذى للشركة، فى حوار لـ(الشروق) على هامش زيارة مصنع «كالونبرج» فى كوبنهاجن الذى أكد المسئولون فيه أنه أكبر مصنع لتصنيع الأنسولين. ويضيف سورينسن أن مصنع «كالونبرج» هو الأكبر من بين مصانع فى 5 دول أخرى هى الولايات المتحدة وفرنسا واليابان والبرازيل والصين، وتتجاوز مساحته المليون متر مربع، ويستحوذ على 50% من إنتاج الأنسولين عالميا، ويعمل 24 ساعة على مدى الـ7 ايام، وينتج يوميا 24 مليون جرعة انسولين لتغطية احتياجات المرضى. وبرغم هذه المساحة فإن عدد العاملين فى المصنع يبلغ 2800 عامل فقط، من أصل 12 الف عامل يعملون فى المصانع الستة على مستوى العالم، حيث يعتمد فى الكثير من المراحل الإنتاجية على استخدام «الروبوت»، تبعا لسورينسن. استثمارات ومصنع جديد لارس أكد أن «نوفو نورديسك» تسعى للتوسع فى السوق المصرية عبر ضخ المزيد من الاستثمارات به، حيث وقعت…
Continue reading …“فايزر” تستثمر 100 مليون جنيه لإضافة خط إنتاج ومستودع العام الجارى
September 10, 2015 Comments Off on “فايزر” تستثمر 100 مليون جنيه لإضافة خط إنتاج ومستودع العام الجارى
1.1 مليار جنيه مبيعات مستهدفة لعام 2015.. وطرح 6 مستحضرات مبتكرة خلال عامين شركات عالمية مهتمة بالاستثمار فى مصر وتعديل منظومة التسعير ضرورى لجذب الاستثمار 33 مليار جنيه مبيعات متوقعة لسوق الدواء 2015.. والشركات الأجنبية تستحوذ على 50% تعتزم شركة فايزر للأدوية إضافة خط إنتاج جديد لتصنيع السوائل، ومستودع تخزينى باستثمارات 100 مليون جنيه خلال الشهور المقبلة. قال عبدالعزيز شاهين، رئيس مجلس إدارة الشركة، إن “فايزر العالمية” وافقت- مؤخراً- على تمويل إنشاء مخزن للأدوية، داخل مصنع الشركة بمصر باستثمارات 70 مليون جنيه، فيما تدرس الشركة الأم- حالياً- إضافة خط إنتاج للسوائل الدوائية بتكلفة تتراوح بين 25 و30 مليون جنيه. وأوضح شاهين لـ”البورصة”، إن المستودع التخزينى يحقق اكتفاء ذاتياً للطاقة التخزينية لمستحضرات الشركة خلال الفترة المقبلة، ويوفر عليها تكاليف التخزين لدى الغير، وتوقع انتهاء تجهيز المخزن خلال الشهور القليلة المقبلة. ورفعت “فايزر مصر” رأسمالها بقيمة تتجاوز 100 مليون دولار يناير الماضى بهدف تمويل الاستثمارات الجديدة، وتوفير التمويلات اللازمة حال التوسع فى المستقبل، حسب شاهين. وتمتلك “فايزر” نحو 70 مستحضراً دوائياً فى السوق المصرى، وتستهدف طرح نحو 6 مستحضرات مبتكرة، لعلاج سرطان الثدى وسيولة الدم والأورام والأمراض النفسية بالسوق المصرية خلال عامين. وتستهدف الشركة تحقيق مبيعات بقيمة 1.1 مليار جنيه خلال العام الجاري، مقابل مليار العام الماضى بنمو 10%، وفقاً لشاهين، الذى…
Continue reading …Novartis to acquire GSK’s multiple sclerosis drug in deal that could surpass $1B
August 24, 2015 Comments Off on Novartis to acquire GSK’s multiple sclerosis drug in deal that could surpass $1B
Novartis AG agreed to buy the rights to an experimental multiple sclerosis drug from GlaxoSmithKline Plc for as much as $1 billion as it races to catch up with a competing treatment being developed by Roche Holding AG. Novartis will pay $300 million upfront to Glaxo for ofatumumab, followed by another $200 million after the start of late-stage clinical trials, the Basel, Switzerland-based company said in a statement on Friday. Novartis may pay as much as $534 million more if certain goals are met during the drug’s development. The drug would compete with Roche’s experimental medicine ocrelizumab, which succeeded in reducing the relapses and disability progression associated with multiple sclerosis in two late-stage studies announced in June. That treatment may reach the market in 2017, while the Novartis drug would be at least two years behind, said Fabian Wenner, an analyst at Kepler Cheuvreux in Zurich. “It’s a joke,” Wenner said by phone. “Patients either want better convenience than the old drugs or they want better efficacy, and ofatumumab is offering neither of those things. The chances of this being successful in MS and generating any sales are zero in my view.” Novartis fell 3 percent to 94.55 euros as…
Continue reading …Amgen Announces Appointment Of Fred Hassan To Board Of Directors
August 1, 2015 Comments Off on Amgen Announces Appointment Of Fred Hassan To Board Of Directors
THOUSAND OAKS, Calif., July 30, 2015 /PRNewswire/ — Amgen (NASDAQ: AMGN) today announced the appointment of Fred Hassan, partner and managing director of Warburg Pincus LLC, to the Amgen Board of Directors. “We are pleased to welcome Fred Hassan and the deep, global experience he brings in the biopharmaceuticals sector to the Amgen Board,” said Robert A. Bradway, chairman and chief executive officer of Amgen. “Fred’s breadth of operational expertise and commitment to innovation will serve Amgen well.” Mr. Hassan has been Partner and Managing Director at Warburg Pincus LLC, a global private equity investment institution, since 2011 and, prior to that, served as Senior Advisor from 2009 to 2010. Mr. Hassan was Chairman of the Board and Chief Executive Officer of Schering-Plough Corporation from 2003 to 2009. Prior to this, Mr. Hassan was Chairman, President and Chief Executive Officer of Pharmacia Corporation, from 2001 to 2003. Before assuming these roles, he had served as President and Chief Executive Officer of Pharmacia Corporation from its creation in 2000 as a result of the merger of Pharmacia & Upjohn, Inc. with Monsanto Company. He was President and Chief Executive Officer of Pharmacia & Upjohn, Inc. beginning in 1997. Mr. Hassan previously held senior…
Continue reading …Hikma to buy Boehringer Ingelheim’s US specialty generics unit for $2.65 billion
July 29, 2015 Comments Off on Hikma to buy Boehringer Ingelheim’s US specialty generics unit for $2.65 billion
Hikma Pharmaceuticals said Tuesday that it agreed to acquire Boehringer Ingelheim’s US specialty generics company Roxane Laboratories under a cash and stock deal valued at approximately $2.65 billion. Hikma noted that the transaction includes around $1.2 billion in cash, while the company will issue 40 million new shares to Boehringer Ingelheim priced at 23.50 pounds ($36.50) each, representing nearly 17 percent of its issued share capital. Hikma CEO Said Darwazah remarked “this transaction has significant strategic value for us, transforming our position and scale in the US generics market.” The company suggested that the deal will make it the sixth biggest US generics provider. The executive added that Roxane’s portfolio “will create a platform for sustainable long-term growth,” while the transaction will also “significantly” expand Hikma’s manufacturing capacity and technological capabilities. Meanwhile, Michael Raya, CEO of Hikma’s US subsidiary West-Ward, said “Roxane is highly complementary to our existing US business.” Raya remarked “we are a market leader in generic injectables. The acquisition of Roxane will significantly strengthen our non-injectables business.” The deal also includes cash payments of up to $125 million from Hikma, subject to the achievement of certain performance milestones. According to Hikma, Roxane is expected to achieve revenue…
Continue reading …Novartis’ new heart failure medicine LCZ696, now called Entresto(TM), approved by FDA to reduce risk of cardiovascular death and heart failure hospitalization
July 9, 2015 Comments Off on Novartis’ new heart failure medicine LCZ696, now called Entresto(TM), approved by FDA to reduce risk of cardiovascular death and heart failure hospitalization
Novartis announced today that the US Food and Drug Administration (FDA) has approved Entresto(TM) (sacubitril/valsartan) tablets, previously known as LCZ696, for the treatment of heart failure with reduced ejection fraction. Entresto will be available on prescription for patients whose condition is classified NYHA class II-IV, indicated to reduce the risk of cardiovascular death and heart failure hospitalization. It is usually administered in conjunction with other heart failure therapies, in place of an ACE inhibitor or other angiotensin receptor blocker. “Despite the uncertainty and high financial risk we designed the world’s largest heart failure trial to compare Entresto to the previous gold standard. As a result millions of people diagnosed with reduced ejection fraction heart failure now have a much greater opportunity to live longer and stay out of hospital,” said David Epstein, Division Head, Novartis Pharmaceuticals. “We recognize our responsibility to ensure Entresto reaches US patients and prescribers as soon as possible and will begin shipping in the US in the coming week.” The FDA’s decision is based on results from the 8,442-patient PARADIGM-HF study which was stopped early when it was shown Entresto significantly reduced the risk of cardiovascular death versus ACE-inhibitor enalapril. At the end of the study,…
Continue reading …Novartis to test new pricing model with heart failure drug
July 2, 2015 Comments Off on Novartis to test new pricing model with heart failure drug
Novartis plans to test a novel pricing model with some customers when it launches its keenly awaited new heart failure drug Entresto, the Swiss company’s head of pharmaceuticals said on Tuesday. Entresto, also known as LCZ696, is the first new drug in decades for helping patients whose lives are in danger because their hearts cannot pump blood efficiently. As a result, it is widely expected to generate billions of dollars in annual sales. How the product should be priced, however, is a dilemma for Novartis, since the company wants to reach as many patients as possible and it knows it will be competing with very cheap – though less effective – older medicines. David Epstein said he was talking to several healthcare customers about a system under which they would get the drug at a discount but then pay Novartis more if, as expected, it successfully reduces the need for costly hospital visits. “We are beginning to share the risk,” he said in an interview. “When you buy other goods that don’t work you either take them back or get your money back. Our industry is a bit unique because historically if the drug doesn’t work it still gets paid…
Continue reading …GSK sells two vaccines to Pfizer to ease competition concerns
June 25, 2015 Comments Off on GSK sells two vaccines to Pfizer to ease competition concerns
GlaxoSmithKline has agreed to sell two meningitis vaccines to Pfizer to satisfy antitrust concerns after its recent acquisition of vaccines business of Novartis. The British drugmaker said on Monday that the total consideration for the sale of its Nimenrix and Mencevax products to Pfizer’s Irish unit, including some deferred payments, was 115 million euros ($131 million). GSK agreed to sell the two older vaccines, which are marketed outside the United States and had combined sales last year of 34 million pounds ($54 million), to meet concerns raised by the European Commission and other antitrust regulators. Meningitis vaccines were an issue for competition authorities because GSK acquired two rival products from Novartis, known as Menveo and Bexsero. The sale to Pfizer is expected to be completed before the end of the year. 25 – June – 2015
Continue reading …The top 15 pharma companies by 2014 revenue
March 20, 2015 Comments Off on The top 15 pharma companies by 2014 revenue
The names at the very top of our yearly list of biggest pharma companies are no surprise. Johnson & Johnson ($JNJ), Novartis ($NVS), Roche ($RHHBY) and Pfizer ($PFE) have headed up the rankings for a couple of years now, albeit in varying order. But toward the bottom of the top 10, there’s been a changing of the guard. For the first time, a biotech company has nudged aside one of the biggest Big Pharma names. Eli Lilly & Co. ($LLY), which hung onto 10th place last year by its fingernails, is now out of the top 10, replaced by Gilead Sciences ($GILD) and its skyrocketing sales. Fueled by its brand-new blockbuster hepatitis C drug, Sovaldi, Gilead revenue more than doubled last year, to $24.5 billion from $10.8 billion. The rest of the top 10 are the usual suspects–Sanofi ($SNY), Merck ($MRK), GlaxoSmithKline ($GSK), AstraZeneca ($AZN) and Bayer HealthCare. They’re in slightly different slots for 2014, but all present and accounted for. But Gilead wasn’t the only Big Biotech to make a major leap last year–and Eli Lilly fell farther down the list than one might think. So, to show all the action in the $20 billion neighborhood, we at FiercePharma…
Continue reading …الشركات والأدوية الأكثر مبيعا فى مصر لعام 2014 بحجم 27 مليار جنيه
March 9, 2015 Comments Off on الشركات والأدوية الأكثر مبيعا فى مصر لعام 2014 بحجم 27 مليار جنيه
اعلنت شركة المعلومات الدوليه IMS عن إحصاء كامل بسوق صناعة الدواء في مصر للشركات العامله في مصر عن عام 2014،وأشار التقرير التى أصدرته الشركة الانجليزية الاسبوع الماضى إلى أن المبيعات الخاصة بالدواء لسنة 2014 للقطاع الخاص بلغت 27 مليارا و461 مليونا و251 ألف جنيه، أي 3 مليار و950 مليون ، و771 دولار بزيادة قدرها 14% عن العام السابق 10% بالجنيه المصري. وشهدت نسبة النمو بالدولار 10% حيث انخفضت عن العام السابق بنسبة 3%، مؤكدا أن السوق الدوائي شهد تراجعا حادًا بالدولار نظرا لارتفاع أسعار الصرف بنسبة 9%. وأعلنت الشركة قائمة أعلي 20 شركة أدوية حققت مبيعات في السوق، وهى شركة نوفارتس وبلغ حجم مبيعاتها 2.397 مليار جنيه، وبلغت عدد العبوات المباعه 149.226 مليون عبوة. واشار التقرير ايضا الى أن الدواء الأكثر مبيعا كان عقار “أوجمنتين Augmentin ، مضاد حيوى لشركة جلاكسو، وبلغت مبيعاته 379 مليونا و636 ألف جنيه بإجمالي عدد عبوات 8 ملايين و517 ألف عبوة، يليه حقن سيفوتاكس «CEEOTAX Vial» مضاد حيوى من انتاج شركة ايبيكو بإجمالي 263 مليونا و428 ألف جنيه، يليه اقراص كاتافلام Cataflam لشركة نوفارتس بإجمالي مبيعات 246 مليون و322 ألف جنيه. واضح التقرير أن سوق المنشطات الجنسية شهد زيادة ونسبة نمو عن العام السابق والأسبق بنسبه 11%، موضحا أن الأصناف الرسمية 8 منتجات فقط المسجلة للمنشطات، حيث بلغت 639 مليونا 361 ألف…
Continue reading …Sanofi names Bayer’s Olivier Brandicourt as new CEO
February 20, 2015 Comments Off on Sanofi names Bayer’s Olivier Brandicourt as new CEO
Sanofi on Thursday announced that its board of directors unanimously appointed Olivier Brandicourt as its new CEO. Brandicourt, who has been chairman and CEO of Bayer’s healthcare division since November 2013, will assume the new position effective April 2. Chairman Serge Weinberg, who said that he was “very pleased” with Brandicourt’s appointment, commented “Sanofi undertook a rigorous selection process to identify the right person to lead Sanofi forward at an important time for our company.” People with knowledge of the matter previously stated that Brandicourt had emerged as the leading candidate to become Sanofi’s CEO, with sources recently disclosing that the Bayer executive could be tabbed as the French drugmaker’s chief executive as soon as this week. Brandicourt will replace former CEO Christopher Viehbacher, who was ousted in October last year over clashes with the board. Commenting on the news, Natixis analyst Philippe Lanone remarked “the nomination of a new CEO would lift some of the uncertainty that has been weighing on shares over the past few months. Brandicourt’s appointment comes as Sanofi struggles with pricing pressure in the diabetes market in the US, with the company announcing earlier this month that revenue for its diabetes division will be flat…
Continue reading …توقيع اتفاقية شراكة ترويجية بين شركتي «جانسن» و«رياض فارما»
February 15, 2015 Comments Off on توقيع اتفاقية شراكة ترويجية بين شركتي «جانسن» و«رياض فارما»
تتويجاً للتعاون الذي أعقب توقيع مذكرة التفاهم بينهما، وقعت شركة جانسن البلجيكية وشركة المنتجات الطبية والتجميلية (رياض فارما) اتفاقية شراكة ترويجية في المملكة لمدة 5 سنوات. ومثل شركة جانسن في مراسم التوقيع إيان فاندر جوتن المشرف العام لشركة جانسن (لمجلس التعاون الخليجي)، فيما مثل شركة رياض فارما الدكتور وليد أمين الكيالي المدير التنفيذي لشركة المنتجات الطبية والتجميلية (رياض فارما). وعلى هامش التوقيع ثمّن الدكتور وليد الكيالي هذه الاتفاقية، منوهاً بأنها تهدف إلى التعاون في تصنيع وتسويق بعض المنتجات الصيدلانية لشركة جانسن البلجيكية في مصنع (رياض فارما) السعودي، مضيفا “سوف ينعكس ذلك في تقوية العلاقات السعودية البلجيكية في جميع المجالات العلمية والاقتصادية، إضافة إلى نقل تقنية الصناعة الدوائية وتوطينها في المملكة ودعم الاكتفاء الذاتي للمنتجات الدوائية والارتقاء بمستوى الصناعات الصيدلانية الوطنية ووصولها إلى مستويات الجودة العالمية”، مشدداً على أن ذلك سيصب في مصلحة المريض أولاً وآخراً، وسوف يساهم في توفير الدواء بصورة دائمة، كما أنه يلبي عدة أهداف تتعلق بالشروط الصحية للدواء من خلال تفادي مؤثرات عمليات النقل والتقلبات الجوية وما ينجم عنها من تأثير سلبي في كفاءة المنتج وحيويته. من جهته ثمّن إيان فاندر جوتن هذه الاتفاقية، معرباً عن ثقته بشركة رياض فارما وما تملكه من قدرات وخبرات عريقة في المملكة ما جعلها محط ثقة شركة جانسن لاختيارها شريكاً في…
Continue reading …Pfizer to buy injectable drugmaker Hospira in $17-billion deal
February 5, 2015 Comments Off on Pfizer to buy injectable drugmaker Hospira in $17-billion deal
Pfizer entered a definitive merger agreement to acquire Hospira for $90 per share in cash, or a total enterprise value of approximately $17 billion, the companies reported Thursday. The price represents a premium of 39 percent to Hospira stock’s close on February 4. “The proposed acquisition of Hospira demonstrates our commitment to prudently deploy capital to create shareholder value,” commented Pfizer CEO Ian Read. The executive added that “Hospira’s business aligns well with our new commercial structure and is an excellent strategic fit for our Global Established Pharmaceutical business, which will benefit from a significantly enhanced product portfolio in growing markets.” Pfizer noted that the deal will add Hospira’s generic sterile injectables product line, including acute care and oncology injectables, as well as its biosimilars portfolio, creating a leading global sterile injectables business.” Pfizer added that the combination also reinforces the strategy of its Global Established Pharmaceutical unit to create a broad portfolio of biosimilars in its therapeutic areas of strength, including a number already marketed by Hospira. According to Pfizer, it will use its resources “to significantly expand” the reach of Hospira’s products, which it said are currently distributed mainly in the US, to Europe and key emerging markets….
Continue reading …The highly recommended: International Guidance for MBA distance learning
January 17, 2015 Comments Off on The highly recommended: International Guidance for MBA distance learning
Click here to download the guide: International Guidance for MBA distance learning – – –
Continue reading …These Are The Drugs Doctors Are Paid The Most To Promote
January 11, 2015 Comments Off on These Are The Drugs Doctors Are Paid The Most To Promote
Companies pay doctors millions of dollars to promote not their most innovative or effective drugs, but some of their most unremarkable. In the last five months of 2013, drug makers spent almost $20 million trying to convince physicians and teaching hospitals to give their freshly-patented drugs to patients, but many of them are near-copies of existing drugs that treat the same conditions. A hefty portion are also available as generics, chemically identical copies that work just as well at a fraction of the price. And still others have serious side effects that only became apparent after they were approved by the FDA. That’s all according to a thorough analysis from ProPublica’s Charles Ornstein and Ryann Grochowski Jones, who combed through federal data on payments made by pharmaceutical and medical device companies to doctors and teaching hospitals and released publicly for the first time last fall under the Affordable Care Act. That data is now available (in a semi-understandable form) on the government website Open Payments. So why are companies paying so much to try to get doctors to prescribe their products? In a word: competition. In ProPublica’s list of the top 20 drugs that companies are paying the most to…
Continue reading …FDA Approves Novo Nordisk’s Saxenda as a Treatment for Chronic Weight Management
December 27, 2014 Comments Off on FDA Approves Novo Nordisk’s Saxenda as a Treatment for Chronic Weight Management
FDA approved Novo Nordisk’s Saxenda (liraglutide injection) as a treatment option for weight management, to be taken in addition to a reduced-calorie diet and physical activity. The drug is approved for use in people with a body mass index (BMI) greater than 30 (considered obesity), or greater than 27 (considered overweight) with at least one additional weight-related condition (e.g. type 2 diabetes, high cholesterol, high blood pressure, etc.). Saxenda is expected to launch in the first half of 2015; there is no pricing information at this time. Saxenda is a once-daily injected GLP-1 agonist that will come in a pen offering up to a 3.0 mg dose. As background, the drug’s active ingredient, liraglutide, is already available in the type 2 diabetes drug Victoza at a maximum dose of 1.8 mg. While Victoza causes a bit of weight loss in many patients, the larger dose of liraglutide in Saxenda helps patients lose much more weight. On average, people using Saxenda in clinical trials lost about 5-7% of their body weight, though more than one in four patients lost over 10% of their body weight. Saxenda was a complicated drug to approve, given that its use for obesity at a 3.0…
Continue reading …
We recently announced a major three-part inter-conditional transaction with Novartis AG involving its Consumer Healthcare, Vaccines and Oncology businesses. More details can be found below. We have released a circular to shareholders and notice of general meeting relating to the transaction. GSK held a General Meeting to approve the proposed major transaction with Novartis AG on 18 December 2014. The proposed transaction would substantially strengthen two of our core businesses in vaccines and consumer healthcare and creates significant new options to increase value for shareholders. It is the most significant transaction for the Company since the creation of GSK in 2000 and is a major step towards fulfilling the Company’s strategy of creating a simpler, stronger and more balanced platform for long-term growth. 25 – December -2014
Continue reading …
عملاق صناعة الدواء في شمال أفريقيا يستعد لدخول سوق السعوديه يبتظر السوق السعودي خلال الايام القليله القادمه انطلاق عملاق صناعة الادويه في شمال أفريقيا ( شركة كووبر للادويه ) ,حيث تعتبر شركة كووبر الاعلى مبيعا في شمال أفريقيا حيث يصل حجم مبيعاتها 260 مليون دولار سنويا. تضع شركة كووبرالابحاث و التطوير ضمن أولوياتها كما أن شركة كووبر لديها شراكات مع أكثر من 20 شركة عالميه مثل أبوت , فايزر , نيوتريشيا و جونسون أند جونسون و تتمثل هذه الشراكات في الحصول على تراخيص التصنيع و التسويق و التوزيع. كما أن قسم الموارد البشريه لدى شركة كووبر يعتمد على التدريب المستمر للموظفين و يسعى الى خلق بيئة عمل متكافئه و ناجحه. للمزيد عن شركة كووبر www.cooperpharma.ma/fr للصيادله في المملكه , فرصتك للانضمام الى فريق عمل كووبر كمندوب دعايه طبيه أو مدير منتجات ( المقابلات الشخصيه خلال أيام قليله ) http://pcm.me/vacancy/cooper-pharma-saudi-arabia
Continue reading …A Note to The Pharmaceutical Industry Upon The Re-Election Of Barack Obama
November 13, 2012 Comments Off on A Note to The Pharmaceutical Industry Upon The Re-Election Of Barack Obama
I’ve covered the drug industry for 12 years, long enough to know that it is no monolith but a collection of hardworking scientists, marketers, and business people, most trying to make a living and make a difference. For a few of you, your jobs are just business, but the vast majority of you do what you do in the hopes of advancing science and helping patients. No single political position accounts for the hundreds of thousands of people working at drug companies, but I know some executives were hoping that Mitt Romney would win. That would have meant that Obama’s rethink of the health insurance system would have been re-thunk yet again, and it might have meant that a lot of pro-pharma ideas on the right, including faster, easier drug approvals and fewer pressures on pricing, would come to pass. But some of this political thinking is inertia, because for decades, on issues like the roll-out of generic drugs, the regulatory climate, and strong protections for intellectual property, drug companies were clearly better off with the pro-business Republican party. It was no accident that Donald Rumsfeld, one of the most prominent Republicans of the past decade, was a former drug…
Continue reading …كندا تنزع براءة الاختراع عن عقار الفياغرا
November 10, 2012 Comments Off on كندا تنزع براءة الاختراع عن عقار الفياغرا
ذكرت شركة “فايزر” للأدوية، الخميس، أنها تتوقع أن تبدأ شركات الأدوية في إنتاج النسخ الخاصة بها من عقار الفياغرا، الذي يعالج الضعف الجنسي بعدما قضت المحكمة العليا في كندا ببطلان براءة الاختراع الخاصة بالعقار. وأيدت المحكمة العليا ادعاء من شركة “تيفا” للصناعات الدوائية الإسرائيلية بأن طلب فايزر الخاص ببراءة الاختراع لم يتضمن معلومات كافية عن المادة الفعالة في الفياغرا. وأفادت فايزر في بيان أرسل بالبريد الإلكتروني، أنها تتوقع مواجهة منافسة من الأدوية غير المحمية ببراءة الاختراع في كندا قريباً. وأعربت الشركة عن خيبة أملها فيما يتعلق بالحكم. وكان من المقرر انتهاء فترة براءة الاختراع لعقار الفياغرا في كندا عام 2014.
Continue reading …سويكال – شركه واعده جديده تقتحم سوق الدواء المصري
October 20, 2012 Comments Off on سويكال – شركه واعده جديده تقتحم سوق الدواء المصري
في خطوه جريئه من شأنها زيادة المنافسه في سوق الدواء المصري , ليس فقط في جودة المنتجات الدوائيه و لكن كبيئه أفضل للموظفين , سيشهد سوق الدواء المصري خلال الأيام القليله القادمه انطلاقة عملاق الدواء الجديد “شركة سويكال للصناعات الدوائيه” شركة سويكال هى شركة مساهمه مصريه برأس مال مصري مرخص به يقترب من نصف مليار جنيه , تم أنشائها من أجل ثلاثة أهداف رئيسيه: 1- انتاج منتجات دوائيه ذات جوده عاليه وبأسعار مناسبه 2- دعم الصادرات المصريه من خلال تصدير المنتجات الى أسواق الشرق الأوسط و دول شرق أوروبا وأفريقيا 3- بناء صرح صناعى طبقا للمواصفات القياسيه العالميه مثل FDA و EMEA وكذلك معامل للتطوير و الأبحاث وقد أرست شركة “سويكال للأدويه” استراتيجيه تعتمد على إستقطاب أفضل كفاءات من الموظفين العاملين بقطاع الأدويه و تمكينهم ببيئه عمل راقيه تعتمد على التدريب و التطوير المستمر للمهارات الشخصيه و العمليه , و يأتي ذلك إنطلاقا من إيمان شركة “سويكال” أن المنافسه الحقيقيه داخل السوق مبنيه على بيئة عمل قويه. وفي تصريح خاص للفارماسيستس كوفي: صرح الدكتور غريب عامر مدير عام الشركة أن شركة “سويكال” تهدف للوصول لحجم مبيعات 400 مليون جنيه بحلول عام 2017 و سيتم الانتهاء من الصرح الصناعى ومعمل التطوير و الأبحاث و بدأ العمل به في عام 2016…
Continue reading …